In marine aquarium science there are many issues that have been passionately debated for decades. Two of the most common issues involve the questions of 1) which is ‘better’ – natural seawater or synthetic sea salts, and 2) trace elements in synthetic sea salts: good or bad? Unfortunately, there is little real science to support any side of the arguments for these topics.
For many people it just seems like common sense to think that natural seawater is better than any synthetic sea salt because, of course, that’s where the animals live. But that thinking assumes that the natural seawater a hobbyist has access to IS the water the animals live in. For most aquarists, even those living on the coast, that is plainly not the case. Furthermore, when it comes to trace elements or heavy metals once again it just “seems” to be right that natural seawater would be the choice. One can look in scientific books and get values for trace elements in seawater, which are very low, which would support the contention that natural seawater is the best choice. But what is the truth?
How much real data is there for us to discuss and develop an informed opinion? In early 2003, an article was published which reflected poorly on the two most popular sea salts on the market; Instant Ocean® and Coralife® (Shimek 2003). The central thesis of this article was that these salts contained huge amounts of many trace elements and so would be toxic in a marine aquarium. “Proof” of this was offered by the author in the way of the results of a bioassay test on these salts and a few others. In this test, a majority of the sea urchin larvae died in the Instant Ocean and Coralife treatments compared to the other treatments. On the surface, it seemed like a pretty open and shut case.
However, the great thing about science is that others can repeat, expand and comment about published work. Over the next several months, my research group will present the results of studies we have done to duplicate the experiment of Shimek. The work will be presented in three parts:
Part 1: A Chemical Analysis of Trace Elements in Synthetic Sea Salts and Natural Seawater.
Part 2: The Toxicity of Synthetic Sea Salts and Natural Seawater to the Development of White Sea Urchin (Lytichinus pictus) larvae, and
Part 3: It is really a “Bad Beginning” or was it just Bad Science: A Rebuttal to Shimek 2003.
The goal of this series of papers is to contribute some real data to the discussion of natural seawater, synthetic sea salt and trace elements. I don’t contend that these papers offer the definitive answer. On the contrary, I believe they offer a foundation on which other studies can build. However, I do think they offer a format for future studies in that they present real data instead of conjecture. I hope everyone will find the papers thought provoking and interesting.
Sincerely,
Dr. Timothy A. Hovanec
- Shimek, R. 2003 The Toxicity of Some Freshly Mixed Artificial Sea Water: A Bad Beginning for A Reef Aquarium. www.reefkeeping.com March 2003.
- In the interest of fair disclosure, it should be pointed out that Dr. Hovanec is the Chief Science Officer for Marineland which makes Instant Ocean synthetic sea salts.
Abstract
Eight commercially available synthetic sea salts (SSS), Instant Ocean (IO), BioSea Marine Mix (BSMM), Crystal Seas Marine Mix Bioassay (CSMMB), Coralife (CL), Tropic Marin (TM), Reef Crystals (RC), Red Sea (RS) and Oceanic Sea Salt (OSS), and two natural seawater samples Catalina Water Company (CWC) and unfiltered seawater (NSW-M) collected from Malibu State Beach, CA, were analyzed via ICP-MS for eleven trace elements and by ICP-MS/DRC for four other trace elements. The majority of the SSS examined had trace element concentrations at values equal to or below those for natural seawater. The trace elements beryllium, cobalt and copper were below the limit of detection for all samples. Silver and Thallium were detected only in OSS. CSMMB was the only SSS with a detectable level of aluminum (10 ppb) but NSW had the largest amount (20 ppb). Cadmium was detected in only two samples: TM (0.31 ppb) and BSMM (0.24 ppb). Zinc was found in four samples: CWC with the highest value (21 ppb) followed by RS (5 ppb), TM (4.1 ppb) and CL (2.9 ppb). Vanadium was detected in four SSS (TM, RC, OSS and CSMMB) but the levels were lower than NSW at 2.0 ppb. Trace amounts (<1 ppb) of chromium were found in five SSS (IO, RC, OSS, RS, and CSMMB) but a sixth (BSMM) contained 27 ppb. Lead was found in six SSS (TM, OSS, RS, CL, CSMMB and BSMM) at various levels below 2 ppb, however, CWC had concentration of 39 ppb. The remaining trace elements (antimony, manganese, molybdenum and nickel) were detected in all the samples at levels generally at or below that of NSW-M except for one SSS. BSMM had significantly higher concentrations of all these trace elements: (Sb-3.5 ppb; Mn-135 ppb, Mo-87 ppb and Ni-108 ppb) compared to all other samples tested. In terms of the total amount of trace elements detected, three SSS (TM, IO and RC) formed a group with levels below 33 ppb while another three (OSS, RS and CL) had a total concentration near 40 ppb. These two groups of SSS had lower total levels of trace elements than NSW-M and CWC. Of the remaining SSS, CSMMB (58.21 ppb) was higher than NSW-M (44.68 ppb) but lower than CWC (73.78 ppb). The last SSS, BSMM, had a total trace element concentration of over 361 ppb.
Introduction
Chemical elements in seawater are commonly grouped into three general categories: major, minor and trace. Pilson (1998) defines the major elements, of which there are eleven, as those that occur in concentrations greater than 1 ppm (1 mg/kg), admittedly an arbitrary value (Table 1). The distinction between minor and trace elements is even more arbitrary and different authors will use different concentrations for the dividing line. Morel and Price (2003) define trace as <0.1 µM while Kennish (1994) states that the average concentration of 22 trace elements ranged between 0.05 and 50 µmol/kg.
| Element / Constituent | Concentration (g/kg) | Element / Constituent | Concentration (g/kg) |
|---|---|---|---|
| *For these chemicals there is no evidence that their concentration varies within any of the major ocean water masses (Pilson 1998). | |||
| Sodium (Na+) | 10.781 | Chloride (Cl–) | 19.353 |
| Magnesium (Mg2+)* | 1.284 | Sulfate (SO42-)* | 2.712 |
| Calcium (Ca2+) | 0.4119 | Bicarbonate (HCO3–) | 0.126 |
| Potassium (K+)* | 0.399 | Bromide (Br–)* | 0.0673 |
| Strontium (Sr+) | 0.00794 | Boric Acid (H3BO3) | 0.0257 |
| Fluoride (F–)* | 0.00130 | ||
What is perhaps more important when distinguishing major elements from minor and trace elements is that major elements are almost all conservative while most minor and trace elements are not. Conservative means that the proportion of the element of concern is nearly constant to other conservative elements and to the salinity of the water. This is also know as Marcet’s Principle or more formally the Principle of Constant Composition (Pilson 1998). Most elements in seawater below a concentration of 1 mg/kg, including many trace elements, are not conservative (Pilson 1998). If an element is not considered conservative it may then be classified as recycled and/or scavenged. However, some elements may fall into two of the three classifications above. For instance, depending upon water depth, copper and iron are both recycled and scavenged (Kremling et. al. 1999). These two elements are depleted in surface waters due to the high productivity associated with this zone of maximum sunlight penetration, and so algae growth, but then their concentrations increase with depth as they are recycled from the decay of marine snow. Table 2 lists some common trace elements, their mean concentration in the ocean and classification according to the above discussion points.
| Element | Concentration (ppm) | Distribution | Element | Concentration (ppm) | Distribution |
|---|---|---|---|---|---|
| (adapted from Pilson 1998) | |||||
| Aluminum (Al) | 0.000270 | mid-depth minima | Manganese (Mn) | 0.000027 | depleted at depth |
| Antimony (Sb) | 0.000146 | ? | Molybdenum (Mo) | 0.010 | conservative |
| Beryllium (Be) | 0.0000002 | nutrient, scavenged | Nickel (Ni) | 0.000470 | nutrient |
| Cadmium (Cd) | 0.000079 | nutrient | Silver (Ag) | 0.0000027 | nutrient, complexed |
| Chromium (Cr) | 0.000208 | nutrient | Thallium (Tl) | 0.000012 | conservative |
| Cobalt (Co) | 0.000001 | depleted at surface | Vanadium (V) | 0.002 | slight surface depletion |
| Copper(Cu) | 0.000254 | nutrient, scavenged | Zinc (Zn) | 0.000392 | nutrient |
| Lead (Pb) | 0.000002 | high in surface waters | |||
An important difference between major and minor or trace elements is that higher concentrations for many minor or trace elements may be toxic to much life in the ocean at various trophic levels and/or life stages. This can be especially important when maintaining aquatic life in closed aquatic systems such as aquaria. Paradoxically, however, many of these “toxic” trace elements are also required nutrients. In fact, the argument among marine biologists as to whether primary productivity in the Ocean is phosphorus or nitrogen limited is muted by the findings that trace elements such as iron can play a more important role in controlling productivity.
Another phenomena which must be considered when discussing trace elements in seawater is the interaction of some elements with particles in the water column. For instance, trace elements such Al, Co, Pb and Mn, which are classified as scavenged types, have strong interaction with particles which results in short residence times in the water column as the particles sink into deeper water (Kremling et. al. 1999). Thus, the toxicity potential of these and other elements must take into consideration the ‘state’ of the element in the water column.
Unfortunately, no research exists on the potentially positive or negative effects of low amounts of various trace elements in aquaria. Indeed, for many years several manufacturers of synthetic sea salts, in order to provide a complete formula, added or separately provided trace elements to their basic formula (Anonymous 1985, 1990). In recent years, however, this practice has nearly stopped as it was realized that many trace elements are naturally present in low quantities in the major chemical compounds, such as sodium chloride, magnesium chloride and sodium sulfate, used to make synthetic sea salts.
However, concern still exists among hobbyists as to whether the use of synthetic sea salts versus natural seawater for marine aquaria results in the continued deleterious introduction of high levels of trace elements to the aquarium. In this study, we analyzed the concentration of several trace elements in synthetic sea salts developed for marine aquaria and compared the results to natural seawater from the coast of California to determine if there were significant trace element differences between these two sources of seawater for aquaria.
Materials And Methods
A natural seawater sample was collected from the surf zone of the Pacific Ocean at Malibu State Beach, CA in a pre-cleaned, acid washed bottle. The sample was stored in a cooler and transported to the laboratory. Salinity was determined with a refractometer (Bio-Marine AquaFauna).
All the synthetic sea salts for this study were purchased from commercial sources except Instant Ocean and Reef Crystals which were obtained from stock at Marineland. Enough of each salt was weighed out to obtain a salinity of 33 ppt to match the salinity of the natural seawater sample. Samples were diluted with deionized water. All samples were coded so that the brand would not be apparent to the analysts. Samples, including deionized water, were transported to West Coast Analytical Services, Inc., Santa Fe Spring, CA in a cooler with ice for analysis.
Samples were analyzed for eleven elements (Al, Sb, Be, Cd, Co, Pb, Mn, Mo, Ag, Tl and Zn) via Inductively Coupled Plasma – Mass Spectrometry (ICP-MS) without further treatment. Another four elements (Cr, Cu, Ni and V) were analyzed by ICP-MS/DRC (Dynamic Reaction Cell). All the samples except OSS were analyzed at the same time. OSS was analyzed at a later date due to its unavailability until that time. During both sample runs, quality control samples were run in parallel on the deionized water used to make-up the synthetic seawater and on fortified laboratory blanks.
Results
Table 3 presents the results of the analysis of fifteen trace elements for eight brands of synthetic sea salts (SSS): Instant Ocean (IO), BioSea Marine Mix (BSMM), Crystal Seas Marine Mix Bioassay (CSMMB), Coralife (CL), Tropic Marin (TM), Reef Crystals (RC), Red Sea (RS) and Oceanic Sea Salt (OSS). Also presented in Table 3 are the data for a natural seawater product for use in marine aquaria, Catalina Water Company (CWC), and natural seawater (NSW-M). The detection limit (DL) for each element and mean published values for the concentration of the elements in seawater are also presented (Table 3).
Examining the results by each element shows that for three elements (Be, Co, and Cu) no sample had concentrations above the detection limit (DL). Another two elements (Ag and Tl) were found only in OSS at values close to (Ag) or just under the DL (Tl). Therefore, these five elements are not considered further, leaving ten elements for discussion.
Aluminum (Al) was detected in only two samples: NSW-M at a concentration of 20 ppb and one SSS, CSMMB with a level of 10 ppb (Fig. 1). Four samples were found to contain levels of zinc (Zn) above the detection limit. CWC had a zinc concentration of 21 ppb which was 4 to 7 times greater than the three SSS (TM, RS and CL) found to contain Zn (Table 3) (Fig. 1).
Detectable amounts of lead (Pb) were found in all treatments except NSW-M, IO and RC (Fig. 1). CWC had the highest level of Pb at 39 ppb. CSMMB and OSS were next highest at 1.7 to 1.8 ppb Pb, respectively (Fig. 1). The remaining SSS had Pb values ranging from 0.37 to 0.82 ppb (Fig. 1).
Chromium (Cr) was detected in six of the eight SSS samples ranging from a high of 27 ppb in BSMM to 0.29 ppb in IO (Fig. 1).
All the SSS, except CL, were found to contain antimony but many were at levels near or below NSW-M (0.36 ppb). BSMM had the highest value at 3.5 ppb followed by CSMMB (1.4 ppb) (Fig. 2). Cadmium was found only in TM (0.31 ppb) and BSMM (0.24 ppb). Four SSS samples were also found to contain measurable levels of vanadium though the amounts were less than that for NSW-M which at 2 ppb equaled the published average value for seawater (Table 2).
The remaining elements (Mn, Mo and Ni) were detected in all the samples tested expect Mo was not detected in CWC (Fig. 3). For these three elements, the general pattern is the same. BSMM had levels of each element that were 4 to 32 times higher than the next highest sample. For two of the elements (Mn and Ni) CSMMB was that next highest sample; for Mo the second highest SSS was TM. Of all the trace elements assayed, Mo is the one highest in natural seawater at an average of 10 ppb. The values for NSW-M (13.4 ppb) and CWC (11.4 ppb) were close to the average seawater value. All the SSS tested, except BSMM, contained less than 10 ppb Mo. BSMM had a MO concentration of 87 ppb Mo. TM was closest to the NSW average with a Mo value of 9 ppb.
Determined values for Mn in the SSS ranged from a low of 7.5 ppb in TM to a high of 135 ppb in BBMM with the remaining SSS having values between 22 and 35 ppb. NSW-M was found to contain 8 ppb Mn while Mn was undetectable in CWC.
Nickel was the only trace element, besides Mn and Mo, that was detected in every sample. Nickel was lowest in the OSS sample (0.37 ppb) which is lower than the average of NSW (0.47 ppb) followed by CWC and NSW-M. BSMM had the highest amount of Ni at 108 ppb. CL and CSMMB were the next highest SSS at 3.3-3.4 ppb. The rest of the SSS had Ni values ranging from 1.1 to 2.2 ppb.
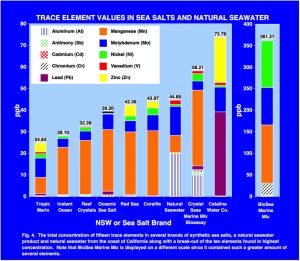
When considering the results from the standpoint of the total amount of trace elements determined, the samples fall roughly into two groups with values below that of the natural seawater sample and then three samples with values higher or much higher than natural seawater (Fig. 4, Table 3). The first group, consisting of TM, IO and RC, had total measured values ranging from 25 to 32.4 ppb and were the lowest tested. The second group, with values ranging from 39 to 44 ppb, included OSS, RS and CL and was close to the value determined for NSW-M of 44.68 ppb. Two SSS and CWC had total trace elements values greater than NSW-M. CSMMB had a total value of 58.21 while BSMM concentration was 361.31 ppb. CWC fell in between these levels with a total value of 73.78 ppb.
| Element | Tropic Marin (TM) | Instant Ocean (IO) | Reef Crystals (RC) | Oceanic (OSS) | Red Sea (RS) | Coralife (CL) |
|---|---|---|---|---|---|---|
| *Detection Limit refers to the lowest possible detection limit within the seawater matrix and analytical procedures of the analyses Values for Natural Seawater are from Pilson (1998) ND – not detected, below detection limit | ||||||
| Aluminum (Al) | ND | ND | ND | ND | ND | ND |
| Antimony (Sb) | 0.24 | 0.61 | 0.62 | 0.18 | 0.18 | ND |
| Beryllium (Be) | ND | ND | ND | ND | ND | ND |
| Cadmium (Cd) | 0.31 | ND | ND | ND | ND | ND |
| Chromium (Cr) | ND | 0.29 | 0.46 | 0.65 | 0.51 | ND |
| Cobalt (Co) | ND | ND | ND | ND | ND | ND |
| Copper (Cu) | ND | ND | ND | ND | ND | ND |
| Lead (Pb) | 0.82 | ND | ND | 1.7 | 0.37 | 0.57 |
| Manganese (Mn) | 7.5 | 22 | 25 | 28.5 | 29 | 30 |
| Molybdenum (Mo) | 9 | 4.1 | 4.2 | 7.1 | 5.1 | 7.2 |
| Nickel (Ni) | 2.0 | 1.1 | 1.8 | 0.37 | 2.2 | 3.3 |
| Silver (Ag) | ND | ND | ND | 0.35 | ND | ND |
| Thallium (Tl) | ND | ND | ND | 0.14 | ND | ND |
| Vanadium (V) | 0.87 | ND | 0.31 | 0.21 | ND | ND |
| Zinc (Zn) | 4.1 | ND | ND | ND | 5 | 2.9 |
| Total Amount (ppb) | 24.84 | 28.10 | 32.39 | 39.20 | 42.36 | 43.97 |
| Element | Natural Seawater (NSW-M) | Crystal Seas Marine Mix Bioassay (CSMMB) | Catalina Water (CWC) | BioSea Marine Mix (BSMM) | Detection Limit | Natural Seawater |
|---|---|---|---|---|---|---|
| *Detection Limit refers to the lowest possible detection limit within the seawater matrix and analytical procedures of the analyses Values for Natural Seawater are from Pilson (1998) ND – not detected, below detection limit | ||||||
| Aluminum (Al) | 20 | 10 | ND | ND | 8 | 0.270 |
| Antimony (Sb) | 0.36 | 1.4 | 0.28 | 3.5 | 0.1 | 0.146 |
| Beryllium (Be) | ND | ND | ND | ND | 0.2 | 0.000 |
| Cadmium (Cd) | ND | ND | ND | 0.24 | 0.2 | 0.079 |
| Chromium (Cr) | ND | 1.0 | ND | 27 | 0.2 | 0.208 |
| Cobalt (Co) | ND | ND | ND | ND | 0.1 | 0.001 |
| Copper (Cu) | ND | ND | ND | ND | 3 | 0.254 |
| Lead (Pb) | ND | 1.81 | 39 | 0.57 | 0.3 | 0.002 |
| Manganese (Mn) | 8 | 35 | ND | 135 | 1 | 0.027 |
| Molybdenum (Mo) | 13.4 | 4.4 | 11.4 | 87 | 0.5 | 10.000 |
| Nickel (Ni) | 0.92 | 3.4 | 0.9 | 108 | 0.3 | 0.470 |
| Silver (Ag) | ND | ND | ND | ND | 0.2 | 0.0027 |
| Thallium (Tl) | ND | ND | ND | ND | 0.2 | 0.012 |
| Vanadium (V) | 2.0 | 1.2 | 1.2 | ND | 0.2 | 2.000 |
| Zinc (Zn) | ND | ND | 21 | ND | 2 | 0.392 |
| Total Amount (ppb) | 44.68 | 58.21 | 73.78 | 361.31 | 13.8638 | |
However, looking at the total amount of the trace elements in a sample does not present a complete picture of analysis. While Tropic Marin (TM) had the lowest total trace element value, it did have substantial amounts of certain elements. Eight of the fifteen trace elements tested were detectable in TM. TM had the highest amount of cadmium (0.31 ppb) which was only detected in one other sample (BSMM – 0.24 ppb). TM was also one of the few sample with detectable amounts of zinc with a value of 4.1 ppb. This zinc value was surpassed only by Red Sea (RS – 5 ppb) and Catalina Water Company (CWC – 21 ppb). The low overall total value for TM is due to the fact that it had lowest Mn value of all the SSS tested, having only roughly a third of the Mn as most of the other SSS (Table 3).
In contrast to Tropic Marin, only five of the fifteen measured elements were detected in Instant Ocean (IO) which had the second lowest total amount of trace elements. No element was found in greatest concentration in IO, and IO had the lowest amount of Mo for all samples. Furthermore, nearly 93% of the total amount of measured trace elements in IO came from just two elements: Mn at 78% and Mo with 14.6%.
The third SSS in the group with the lowest amounts of measured trace elements was Reef Crystals with a value of 32.39 ppb (Table 3). Made by the same company that manufactures IO, but designed to contain certain trace elements and minerals for reef tanks, it is not surprising that RC is close to, but slightly higher than IO, in terms of trace metal concentration. Again, like IO, RC did not contain the highest amount of any trace element for the SSS tested and slightly over 77% of the total trace elements was due to one element (Mn) with another 13% due to Mo.
The second group of SSS, Oceanic Sea Salt (OSS), Red Seas (RS) and Coralife (CL), while having a total amount of the trace elements measured lower than NSW had varying amounts of additional elements when compared to the first group of SSS. For instance, both RS and CL were two of three SSS with measurable levels of zinc and RS has the highest zinc level of all SSS tested (Table 3). OSS had one of the highest amounts of chromium (1.65 ppb) and lead (1.7 ppb) for all SSS and was the only seasalt with detectable levels of silver and thallium. However, OSS did have the lowest amount of Ni for all samples tested.
The two remaining SSS tested, CSMMB and BSMM, had slightly higher and considerably higher total amounts of measured trace elements, respectively. BSMM had a total of 361.31 ppb of trace elements and had the greatest amount of five of the twelve elements detected in all samples (Sb, Cr, Mn, Mo, and Ni). For many of these five elements, the amounts found in BSMM were an order of magnitude higher than the next lowest SSS. CSMMB had the greatest amount of Al of all SSS tested and also had significantly higher concentrations of five other elements (Sb, Cr, Pb, Mn and Ni) compared to the other SSS besides BSMM.
Discussion
The results of this study demonstrate that the vast majority of synthetic sea salts for use in marine aquaria do not have concentrations of the trace elements examined that are substantially greater than that of the natural seawater available to the marine hobbyist. Furthermore, for some trace elements natural seawater had a greater value than some of the synthetic sea salts. Moreover, Catalina Water Company, which is a natural seawater product, had a lead value that was one to two orders of magnitude higher than other samples. It is clear that most synthetic sea salts do not add substantial amounts of trace levels to aquaria when compared to the natural seawater available to the majority of hobbyists.
However, whether this is beneficial or detrimental to the aquarium environment is not known. As previously mentioned, there are no scientific studies applicable to marine aquaria which allow one to say with a high degree of confidence that a certain amount of a particular trace element is absolutely necessary in the marine aquarium or that a constant low level of a certain element may cause a poisoning of the aquarium. This is one area in need of much research.
Conversely, while the results of toxicity studies done on fish and some corals allow one to predict that high concentrations of some trace elements would be detrimental to the aquarium environment, the results of this study show that most synthetic sea salts do not contain such dangerously high levels of trace elements for this to be a concern to the aquarist.
It must be realized that the data herein are only for one sample of each product at one time and subsequent analysis may produce different values. This could be especially acute for any natural seawater product because of biotic and abiotic environment factors such as rain, algae blooms, ship traffic off the near coast, etc. In fact, trace elements in synthetic sea salts would probably vary less over time because manufacturers of these products have more control over the raw materials used to make the product compared to the total lack of control over the natural oceanic environment.
Many of the determined trace element values for the natural seawater samples in this study were much higher than published values (Table 3). However, published values are by necessity generalizations. Actual values will vary due to locale and local factors that have to be taken into consideration. For example, Pilson (1998) noted that lead is high in surface waters most likely due to anthropomorphic reasons. There are many other potential reasons why the determined values in NSW may be much higher than average published values for NSW. First, the actual concentration of any trace element in NSW will vary considerably spatially and temporally. Furthermore, water samples taken in coastal areas and near major metropolitan areas would be expected to have elevated concentrations of many elements due to run-off, winds from the coastland, exposure to polluted air, upwelling, shipping traffic in the ocean channels off the coast, and associated factors. Plus, primary productivity can be very high in coastal areas which can affect the concentration of many trace elements as organisms consume and recycle nutrients, including trace elements, in the course of their natural life cycles. These factors are especially important for trace elements that are nutritive rather than conservative.
Consider that the natural seawater sample (NSW-M) in this study was determined to have the highest amount of Al of all samples with a value of 20 ppb. This value is not as strange as it may first seem and further illustrates the problem with relying on published general values for NSW. Pilson (1998) relates how atmospherically transported dust, rich in clays containing aluminum, may be the cause of the surface enrichment of aluminum. This could certainly be the case with the natural seawater sample in this study which was collect just beyond the surf zone off Malibu State Beach, CA.
The high values of lead and zinc in the natural seawater from Catalina Seawater Company is most likely due to fuel fumes associated with the ship collecting the seawater and the truck used to transport the collected seawater. High surface concentrations of lead in seawater have been linked to atmospherically transported lead mainly from the exhausts of automobiles and trucks (Pilson 1998).
It is perhaps natural that in discussing trace element values in synthetic sea salts authors would want to compare values in the salts with those of natural seawater. However, that view assumes that hobbyists have access to natural seawater with trace element values that match the values in the book or paper the author is referring to. The results of this study show that this is a false assumption. Natural seawater that is available to the hobbyist comes from near shore sources and it should not be surprising that water quality in these oceanic zones differs greatly from that of more remote oceanic areas.
A question in the back of the minds of researchers is – how good are the data? For this study that means how correct is the analysis? There are several ways to check the validity of the data. A seawater reference standard with known values was run along with the samples of this test. The determined values for the reference standard were in close agreement with the known values which tells one that the analytical methods (ICP-MS and ICP-MS/DRC) were sufficiently accurate for this examination. Further support for the validity of the test analytical methods can be found by looking at some of the values for select trace elements for natural seawater. For example, one of elements tested, molybdenum is conservative and has a mean value in the ocean of 10 ppb, with a range of 9.2 to 10.5 (Table 2) (Pilson 1998). The determined values for natural seawater in this study, 11.4 to 13.4 ppb, are in close agreement with these data adding validity to the analytical procedure.
In fact, this study may be the first to analyze natural seawater samples alongside samples of synthetic sea salts. Earlier studies on trace element concentrations in SSS did not include natural seawater samples for reasons unknown. Atkinson and Bingman (1997) analyzed a number of commercial sea salts but did not actually include a seawater sample. Instead they listed values for various elements and ions in seawater.
Atkinson and Bingman (1997) included a number of sea salts in their analysis that were also included in the present study (IO, TM, RC, RS, and CL). In general, their determined values for most trace elements were much higher than the values found in this study. The most likely explanation for these differences is that the analytical method used by Atkinson and Bingman (1997) to determine trace element concentration in the sea salts was not sufficiently precise and was subject to interferences which caused false high readings. Atkinson and Bingman (1997) used Inductively Coupled Plasma emission spectroscopy (ICP) for their analysis. The current study used either ICP-MS or ICP-MS/DRC for the trace element analysis. These methods are more precise than ICP and can yield much better data. ICP-MS/DRC is used specifically to remove interferences associated with the masses of various elements during ICP-MS that can cause false high reading for certain elements such as chromium, copper, nickel and vanadium. Shimek (2002a) reported the analysis of a sample of Instant Ocean made with reverse osmosis/deionized (RO/DI) water for trace elements using ICP scan. His results were more like those of this study; cadmium, chromium, lead, and manganese were below the test detection limits. However, he also failed to include a natural seawater sample for comparison and the ‘average’ natural seawater presented were incorrect (Shimek 2002b).
The chemical analysis of trace elements is not a straightforward task. A trace element at a concentration of 1 µg/kg (1 ppb) in seawater is in a matrix of elements and compounds that total nearly 35,000,000 µg (Pilson 1998). Contamination during sampling, interference between elements, swamping of the signal for one element by another element in much greater concentration plus many other factors combine to make the analysis difficult.
In addition to the technical difficulties, the fact that many of the trace elements have a biological role such that their concentration will vary throughout the water column depending upon plankton uptake and recycling leaves one to conclude that for many non-conservative elements valves will range considerably.
The concern over trace element concentrations in synthetic sea salts is due to the fact that some trace elements, also known as heavy metals, are toxic to marine organisms. However, heavy metal toxicity is a complex phenomenon, especially so in marine environments. The straightforward question of what is the toxic concentration of a specific metal is not easily answered. Furthermore, a dozen or so metals with an atomic mass over 50, including Mn, Fe, Co, Ni, Cu Zn and Cd, have known biologically roles (Morel and Price 2003). In fact, the surface depletion of most trace metals is due to uptake by plankton (Morel and Price 2003). This leads to the situation where the concentration of many trace metals is orders of magnitude greater in the plankton compared to the water in which the plankton live. Trace metals can also be transferred and concentrated in the body of organisms at different trophic levels in the marine food web (Twining and Fisher 2004).
Three other major factors that confound efforts to determine actual toxicity levels of many heavy metals are solubility, speciation and chelation. The analytically determined value of any heavy metal may not equate to the actual amount of the metal available, either for nutritional purposes or toxic effects, in the seawater environment. Pilson (1998) states that it is known that some metals, such as Fe, Co, Cu and Zn, are quite strongly complexed with organic matter in seawater and provides a detailed example, using Cu, of how toxicity is affected by this interaction. Briefly, studies have shown that the toxicity of copper is due to the concentration or activity of the free copper ion in solution. However, it has been calculated that about 90% of the copper ion in seawater (at 15°C and normal pH) is complexed mostly with carbonate and some hydroxyl ions. Taking into account the activity coefficient of the copper ion, Pilson calculates that only 2% of the actual Cu concentration is in the free (toxic) form.
Morel and Price (2003) state that for metals such as Fe, Co, Cu, Zn and Cd the bulk of the dissolved concentration in seawater is present in the nonreactive (nontoxic) form at the ocean surface. The working theory is that this is due to the metals being bound to some strong unknown ligands (or binding molecule) with a small fraction of the metals being colloidal. These putative ligands, or chelators, some of which have been shown to be from biogenic sources are presumed to detoxify the metals and possibly assist with metal transport and sequestration (Morel and Price 2003).
Some metals – Fe, Mn, Cu and Co – have more than one oxidation state in seawater and may further be subjected to a dynamic redox cycle (Morel and Price 2003). This redox cycle is powered by light and promotes charge-transfer reactions in some metal complexes which causes the release of the metal from the oxidized ligand. In fact, dissolved Mn is atypically, compared to many other elements, at maximum concentration at the ocean surface due to the photochemical reduction of Mn (IV) oxides to soluble Mn (II) (Morel and Price 2003).
In summary, trace element testing and toxicity are complex and many factors must be considered besides just a hard to determine analytical value. However, this study demonstrated several important conclusions in regards to comparing synthetic sea salts to natural seawater that should be of major importance to marine aquarists:
- most synthetic sea salts do not have high levels of many trace elements,
- natural seawater is not automatically a safe alternative to synthetic sea salts when it comes to comparing amounts of trace elements in the two solutions,
- past comparisons of synthetic sea salts to natural seawater are flawed because they compared synthetic sea salts to a hypothetical natural seawater that is not available to the majority of marine hobbyists, and
- synthetic sea salts are quite acceptable for long-term use in marine aquaria when considered from the viewpoint of not adding detrimental concentrations of trace elements to the system.
Acknowledgments
We wish to thank Elena Toy, Jennifer Westerlund and Jason Niemans for their assistance with this study.
Correspondence
Corresponding author: Mailing address: Marineland, 6100 Condor Dr., Moorpark, CA 93021. Phone 805.529.1111. Fax 805.529.3030. Email: [email protected]
References
- Anonymous. 1985. Synthetic Sea Salts: Trace Elements, But So Much More. SeaScope Vol 2 Spring 1985 page 1.
- Anonymous. 1990. Trace elements and Sea Salt Mixes. SeaScope Vol 7 Summer 1990 page 1.
- Atkinson, M.J. and C. Bingman. 1997. Elemental composition of commercial seasalts. J. Aquari. & Aqua. Sci. 8:39-43.
- Kennish, M.J. 1994. Practical Handbook of Marine Science, 2nd Ed. CRC Press Boca Raton. 566p.
- Kremling, K., M.O. Andreae, L. Brügmann, C.M.G. van den Berg, A. Prange, M. Schirmacher, F. Koroleff and J. Kuss. 1999. Determination of Trace Elements p. 253-364. In: Methods of Seawater Analysis 3rd ed. K. Graddhoff, K. Kremling and M. Ehrhardt eds. Wiley-VCH Weinheim, Germany.
- Morel, F. M. M. and N. M. Price. 2003. The Biogeochemical Cycles of Trace Metals in the Oceans. Science 300(5621):944-947.
- Pilson, M.E.Q. 1998. An Introduction to the Chemistry of the Sea. Prentice-Hall, Inc. Upper Saddle River, N.J. 431p.
- Shimek, R.L. 2002a. It’s (In) The Water. Reefkeeping Online. www.reefkeeping.com Feb. 2002.
- Shimek, R.L. 2002b. It is Still In The Water. Reefkeeping Online. www.reefkeeping.com Mar. 2002.
- Twining, B. S. and N. S. Fisher. 2004. Trophic transfer of trace metals from protozoa to mesozooplankton. Limnology and Oceanography 49(1):28-39.





0 Comments