Only a few corals can match this coral’s startling contrast of colors, making the Superman Montipora a highly desirable animal for display reef aquaria. As can be expected, the demand for this coral ensures a premium price.
The following article will examine some husbandry techniques for this exotic coral. Since many of us are not particularly interested in how information within this article was obtained, I’ll present the pertinent data first and will save the nitty-gritty technical details and methods for the end of this article.
Again, bear in mind that the details below are from a limited number of specimens, and other Montipora species may bear a resemblance to M. danae (be sure of the coral’s ID!).
Common Name: Superman
Family: Acroporidae
Genus: Montipora
Variously described as these species: Montipora danae (which can be confused withM. verrucosa, M. verruculosus and M. palawanensis. The latter two species are uncommon and restricted to a smaller geographical area than the two former species). Also described in advertisements as M. tuberculosa. The coral in Figure 1 is likely M. danae as its immersed corallites are widely spaced between hillocky coenosteum verrucae (or tuberculae) and partially fused ridges. Identification is based on results obtained by using Veron’s Coral ID software. See Comments below for basis of identification.
Geographical Range: Indian and Pacific Oceans and possibly the Red Sea.
Known Symbiont Types: Symbiodinium species, including Clades C2, C31, C+ and C·.
Light Requirements
Reef hobbyists, as a group, have the correct concept about lighting – it is an important factor in successful reefkeeping and probably the most expensive routine maintenance item in terms of electricity consumption. Yet relatively few own or use any sort of light meter. This is probably due to the fact that so little is known about light requirements of photosynthetic invertebrates. However, there are instruments available to determine how much light is actually required. As this information becomes available, we can arm ourselves with this knowledge and can potentially begin to put our reef aquaria on energy diets. In short, a light meter is a good investment.
At a minimum, hobbyists should have (or have access to) a lux meter with a submersible sensor or, even better, a quantum meter for measuring Photosynthetically Active Radiation (PAR, reported in units of micro Mol photons per square meter per second, or µmol photons·m²·sec, but usually just µmol·m²·sec. Maximum PAR in the tropics at noon on a cloudless day can be as high as 2,200 µmol·m²·sec).
Although either of these instruments is relatively expensive, the price of a quality meter has dropped dramatically over the last decade or so. No longer is it necessary to spend over $1,000 for a PAR meter – quite adequate units are available for ~$250.
I have to wonder what state the hobby would be in if we treated many of the established parameters in the manner many treat light intensity. What if we were advised to maintain either a high or low pH? Alkalinity? Phosphorus? Calcium?
With that said, how does light does a Superman Montipora require? Instead of saying ‘not much’ or ‘a lot’, we can estimate the light requirements via the technique of non-invasive fluorometry. What does that mean? It simply means that we can, with proper instrumentation, observe how energy flows in the photosynthetic process. This energy flow is called the ‘Electron Transport Rate’ (or ETR for short). The higher the ETR, the higher the rate of photosynthesis. Here, we report ‘relative ETR’ (rETR) since absorption of light by photopigments within zooxanthellae was not measured.
It is a common misconception that reef aquaria cannot have too much light. To the contrary, evidence suggests many corals actually photo-saturate (where the rate of photosynthesis – the rETR – does not increase with increasing light intensity. For examples, see Ulstrap et al., 2006, where the SPS coral Pocillopora damicornis becomes photo-inhibited at ~450 µmol·m²·sec).
Photoinhibition is possible. Photoinhibition is the phenomenon of reduced photosynthesis where available light exceeds that needed for maximum photosynthesis. In these cases, zooxanthellae are trying to protect themselves from photo-destruction via dumping excess energy as chlorophyll fluorescence in a process called photochemical quenching. In addition, xanthophyll pigments within zooxanthellae can absorb blue light and dissipate this energy as non-radiant heat that is called non-photochemical quenching. So, what amount of light is sufficient for growth and coloration of the Superman coral?
Figure 2 demonstrates the rate of photosynthesis of one Superman specimen under various light intensities. For those arguing that these numbers represent only a snapshot of one coral’s zooxanthellae after photoacclimation (which, of course, they do), I suggest that they’ve missed the point! The point is not so much that these rETRs are representative of zooxanthellae photosynthetic activity after photoacclimation to a given amount of light – it is the fact that the zooxanthellae have the ability to adapt to this particular amount of light!
If we assume that calcification rates are linked to rates of photosynthesis, then Figure 2 takes on added meaning. Examination of Figure 2 suggests photosynthesis (and hence calcification and growth) would theoretically be about the same at 250 µmol·m²·sec and 500 µmol·m²·sec. However, bleaching of a Superman specimen has been noted at a light intensity of only 300 µmol·m²·sec (and a photoperiod of 11 hours. It seems certain that light – and not temperature, ultraviolet radiation, or any other ‘nasty’ factor – is responsible for the bleaching. This observation certainly deserves further investigation).
The bottom line – analyses suggest these two M. danae specimens and their zooxanthellae seem to prefer lower light conditions, where intensity is only 100 – 200 µmol·m²·sec.
Known Symbionts
Recent ‘fingerprinting’ of zooxanthellae DNA has revealed a large number of sub-species or ‘clades’. There are several known ‘types’ of zooxanthellae found in Montipora danae, including Clade C2 (based on ITS2 analyses, Van Oppen, 2004; 2005), C31 (LaJeunesse, 2004; Okinawa, Japan, 1-10m), C+ and C· (ITS1; Van Oppen, 2004).
Clade C2: This zooxanthellae clade is found largely in Family Acroporidae corals (Acropora, Montipora, among others) including Acropora aspera, Acropora cerealis (GBR, Van Oppen, 2001), Acropora cervicornis (Caribbean 2.0-17.0m, Baker et al., 1997), Acropora cuneata (Van Oppen et al., 2005), Acropora florida, Acropora gemmifera, Acropora intermedia, Acropora longicyathus, Acropora loripes, Acropora millepora, Acropora nastua, Acropora spathulata, Acropora tenuis, Acropora valida (GBR, Van Oppen, 2001),Montipora aequituberculata, Montipora capricornis, Montipora danae, Montipora florida (from Indonesia, Van Oppen, 2005 based on ITS2 fingerprint). Clade C2 has also been reported from Pavona varians (Van Oppen, 2005), Goniastrea rectiformis (Van Oppen, 2005), zooxanthellae collected and cultured from the clam Hippopus (LaJeunesse, 2003), and Pocillopora damicornis (two locations in Taiwan, 0-5.0m, Chen et al., 2005). C2 is believed to have adaptively radiated from Clade C3 (a ‘generalist’ zooxanthella).
It is an interesting notion that corals containing Clade C2 could possibly have the same range of photoacclimation. Photoacclimation in most zooxanthellae is not infinite in range, although it seems certain that some ‘types’ of Symbiodinium have more latitude in adjusting to light intensity than others. This is an important concept – zonation of certain corals has been linked to light intensity (Iglesias-Prieto et al., 2003), though factors such as temperature tolerance and others also play roles.
Clade C31: So far, Clade C31 seems to be most common in Montipora specimens. It has been found in Montipora danae specimens (Okinawa, Japan, 1-10m; LaJeunesse, 2004) as well as Montipora species (western and Central Pacific 2-20m; LaJeunesse et. al., 2003); Montipora capitata (Hawaii, 1-5m, LaJeunesse, 2004) and Montipora patula (Hawaii, 20m, LaJeunesse, 2004). C31 is believed to have evolved from Clade C21. Clade C31 has also been found in Montastraea annularis (Belize, 8m; Warner et al., 2006).
A pulse amplitude modulation (PAM) analysis of photosynthetic capacity of a shallow-water Hawaiian Montipora capitata (believed to contain Clade 31) found onset of photosaturation at ~135 µmol photons·m²·sec (~6,750 lux). Photoinhibition is thought to occur at ~250 µmol photons·m²·sec (Riddle, in press).
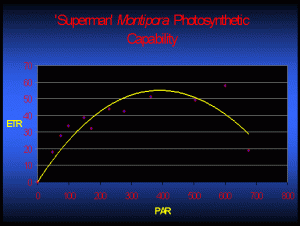
Figure 2. Photosynthetic activity (relative Electron Transport Rate, or rETR shown by the yellow line) of one Superman Montipora specimen. In this case, maximum photosynthesis occurs at a PAR value of ~400 µmol·m²·sec. Increasing light intensity above this point is counter-productive as photoinhibition occurs (as indicated by reduced rETR).
Based on the information about coral species and associated depth presented by researchers (above), it appears that Clade C31 zooxanthellae probably tolerate, if not prefer, lower light intensity.
Clade C+: Isolated from the Pacific stony coral Montipora danae (van Oppen 2004) in combination with Clade C2 (Van Oppen, 2004) and Plesiastrea verispora (Magalon et al., 2007).
Clade C·: This zooxanthella is believed to have co-evolved with Montipora species, but sometimes found in Porites attenuata and Porites cylindrica. Montipora species containing Clade C· include Montipora aequituberculata, M. altasepta, M. angulata, M. cactus, M. capitata, M. crassituberculata, M. danae, M. delicatula, M. digitata, M. gaimardi, M. hispida, M. hoffmeisteri, M. mollis, M. peltiformis, M. spongodes, M. stellata, M. turtlensis, M. undata, and M. verrucosa (van Oppen et al., 2004). This clade is presently known to be distributed from Indonesia southward to the Great Barrier Reef. One has to wonder if this clade has high fidelity to Montipora spp. and is one of those listed in LaJeunesse’s more-or-less concurrent paper (namely Clades C17, C26a, C27, C30, C31, C31a, C31b, C32, C58 and C73). Van Oppen’s IDs are based on ITS1 sequences (while LaJeunesse’s – and many others’-
are based on ITS2 fingerprinting).

Figure 3. Appearance of the Superman Montipora when illuminated by a warm-white fluorescent lamp. The non-fluorescent chromoprotein reflects blue and red light, making the coral appear violet/purple.
Possibilities of ‘Symbiont Shuffling’
Are the clades listed above the only zooxanthellae inhabiting Montipora danae? Probably not. But it is the best information currently available; however, new laboratory techniques will undoubtedly change our view.
Warming of the earth’s oceans has generated much interest in how corals and their zooxanthellae respond to environmental conditions. Researchers are identifying the possibility of ‘symbiont shuffling’ where one zooxanthella clade loses its dominance due to unfavorable conditions (excess light, UV, temperature, etc.) and is replaced by a second clade that finds the new environment as hospitable and thus becomes dominant.
Much of the recent research work involves techniques that cannot detect zooxanthellae populations when they are less than 5-10% of the total population (although Mieog et al., 2007, report a technique with 100-fold increased sensitivity. Future results using this technique should prove quite interesting). Only one case to my knowledge demonstrates two clades living simultaneously in a M. danae specimen (C2 and C+; Van Oppen, 2004).

Figure 4. The same coral in Figure 3. Note the considerable growth that has occurred over 4 months time under ‘low light’ conditions – only 100 µmol·m²·sec, although much of the non-fluorescent purple coloration has disappeared.
Zooxanthellae inhabiting Montipora specimens likely have a high fidelity for their host (and vice versa); In fact some clades are believed to have co-evolved with their host coral. In addition, Montipora larvae acquire their symbionts directly from the parental colony – the eggs are infected by zooxanthellae from the parent – in a process called maternal acquisition (not all corals behave in this manner – many larvae obtain symbionts from the water column). In either case, acroporid corals seem to have a preference for specific zooxanthella clade(s) (van Oppen, 2004).
Coral Pigmentation – What Color Do You Desire?
Coloration of the Superman coral is dependent upon the type of light and here’s why: Analyses of fluorescence have revealed the presence of at least six pigments within the Superman Montipora. The major pigment is found within coral tissues and fluoresces at a peak of 489nm (blue-green). Their reddish color of the polyps is due to fluorescence peaking at 611nm (see Figure 6). Other minor fluorescent pigments are seen at 546, 567, 587, and 617, with possible fluorescent spikes at 491, 520 and 540nm. There is also an unidentified non-fluorescent chromoprotein at can make the coral appear blue or purple (depending upon the spectrum of the illumination source).
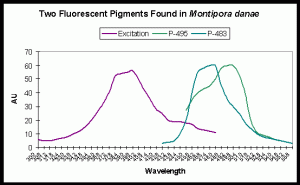
Note that other M. danae specimens can contain different pigments. For instance, Salih et al. (2006) identified two additional fluorescent pigments. Fluorescent emissions are seen at 483 and 495nm. Excitation wavelengths include UV-A, violet and blue wavelengths (see Figure 7).

Figure 5. A closer view of the coral pictured in Figures 3 and 4. A high kelvin lamp will showcase the blue, green and orange fluorescent pigments.
Figure 7 is useful in understanding which wavelengths will make this coral fluoresce in the blue-green range. Although ultraviolet light will induce fluorescence, the maximum excitation wavelength is in the violet portion of the spectrum.
The purple chromoprotein seems to be expressed as a result of light intensity (although other ‘proper’ conditions in aquaria are also required). At light intensity of about 100 µmol·m²·sec, the purple coloration is barely seen, but growth is good (see Figure 4). The bluish chromoprotein can become the apparent dominant pigment at ~175 µmol·m²·sec.
While non-fluorescent coloration seems dependent upon light, the fluorescent orange seen in the polyps and the blue-green ‘body’ pigments (produced by the coral animal and not zooxanthellae) are usually seem except when the light intensity is too high (and the coral bleaches) or when light is too low (and the zooxanthellae/host suffer from light deprivation. Though strictly speculation on my part, the host probably stops producing pigments as an energy-saving measure).
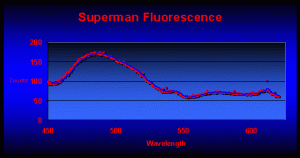
Figure 6. Fluorescent emission of a Superman coral reveals a major peak at 489nm. The reddish polyps fluoresce strongly at 611nm.
Water Motion
This coral’s ‘bumpy’ surface (called tuberculae or verrucae, depending upon their size relative to corallites) indicates this animal requires ‘good’ water flow. These tuberculae act as ‘speed bumps’ to laminar water flow and create turbulence, which is not surprising as these corals are often found on upper reef slopes. Slowing of water velocity by these bumps prevents deformation of polyp shape and allows the coral to feed.
Fortunately, creating sufficient water movement is quite easy with some of the newer propeller pumps and other devices (see Riddle, 2007).
Other Aquarium Conditions
The Superman Montipora fares well when other physical and chemical conditions in an aquarium are within ‘reasonable’ ranges. To avoid potential overheating of coral specimens, water temperature should not exceed 80º F when using metal halide lamps (see Riddle, 2006 for details). Although a link to coloration in aquaria conditions needs to be established, alkalinity and its potential relationship to coral tissue pH (Battad et al., 2007) under lighted conditions could also play a role in coral coloration.
Methods and Materials
Rates of photosynthesis were determined with a Walz ‘Teaching PAM’ Chlorophyll Fluorometer (Effeltrich, Germany) equipped with a fiber optic cord. A fluorometer is basically a ‘photosynthesis meter’ and exploits measurements of chlorophyll fluorescence in order to determine rates of photosynthesis (in terms of ‘relative electron transport rate’ or rETR between Photosystem I and Photosystem II within zooxanthellae. Since pigment absorption of light was not measured, the ETR is considered to be relative). Corals were maintained in total darkness for at least one hour before initial measurements were made in order to allow Photosystem reaction centers to ‘open’. The external actinic light source was a 400-watt Iwasaki 6,500K metal halide lamp that was shielded for ultraviolet radiation by a clear acrylic material. Intensity was adjusted upwards by moving the light source closer to the coral (which was contained in a 3-gallon plastic container). Light intensity (Photosynthetically Active Radiation, or PAR) was measured with an Apogee QMSS quantum meter and a submersible, cosine-corrected sensor. Water motion was provided by a magnetic stirrer and a 3″ stir bar.
After the dark-adaptation period, minimum chlorophyll fluorescence (Fo) was determined with weak light (<1 µmol·m²·sec) generated by the instrument’s internal actinic lamp. Increasing the intensity of the internal actinic lamp to provide a saturating light pulse determined Maximum Fluorescence (Fm). The metal halide lamp was then turned on and allowed to warm up until PAR values stabilized (15-20 minutes). Chlorophyll fluorescence values were determined and the light intensity increased. The coral’s zooxanthellae were allowed to adjust to the new light intensity for 15 minutes and another chlorophyll fluorescence measurement was made. The PAM meter calculated Photosynthetic Yield, which was simply multiplied by the appropriate PAR measurement in order to estimate the ‘relative Electron Transport Rate’ or rETR. This is a valid method for observing photosynthetic trends.
Fluorescence was determined with an Ocean Optics USB-2000-FL fiber optic spectrometer (Dunedin, Florida) using an 18-watt black light (maximum emission at 365nm) as the excitation source. Light was collected with a cosine-corrected CC-3 lens.
Photographs were taken with a Canon Rebel XTi digital camera (10.1 megapixels) equipped with two stacked teleconverters and a 60mm macro lens.
Comments on Basis of Identification
Veron (2000) states: “The identification of aquarium-raised corals is a difficult and sometimes impossible task… They may also assume growth forms which are sometimes seen in the wild, but usually only in deep water that may be unusual for the species.” With that said, hobbyists are cautioned that information contained within this article was obtained from two aquarium-grown specimens, and, although best efforts have been made to identify these animals, limitations of the software lend a degree of uncertainly as to the exact coral species.
In an effort to identify these corals, twenty-seven identification points, including those observed from two living and one skeletal remains, were entered into Veron’s Coral ID software. These observations for data entry were made from examinations of enlarged photographs of living corals and microscopic examinations of a skeleton.
To properly identify a coral, its geographic origin should be known, and in these cases, the collection point(s) is not known. Assuming these corals originated from the central Indo-Pacific Ocean (including Indonesia, Philippines and Solomon Islands), the software determined identity as Montipora danae or M. undata, with the software’s ‘Best Bet’ option signaling M. danae as the proper identification.
While changing the location data point (and retaining all others), the identification consistently included M. danae. For instance, entering Oceanic West Pacific (including Fiji) the software also returned two possibilities – Montipora danae and M. undata. Similar results were listed for SW Indian Ocean. Central Pacific (including Tonga) suggested only one possibility – M. danae.

Figure 8. Distinctly different skeletal growth forms within millimeters of each other. To the right, linear elaborations typical of many Montipora specimens are dominant. To the left, the skeleton flattens to a rather featureless, flattened growth pattern.
These are the other identification criteria (those items in italics were determined by microscopic exams):
- Multiple mouths and corallites
- Attached to the substrate (as opposed to free-living)
- Hemispherical, sub-massive or encrusting growth forms
- Branching absent
- Calice width less than 1mm
- Corallite centers distinct
- Corallites separate individuals
- Immersed corallites
- Neither axial nor central corallite
- Corallites widely spaced (more than twice the diameter of the corallite opening)
- Tentacles expanded by day
- Tentacle length <10mm
- Partial skeletal masking
- Daytime tissue projection <1mm
- Extra-thecal skeleton present
- Extra-thecal surface perforated
- Linear elaborations present
- Linear elaborations greater than calice diameter
- Columella absent
- Costae absent
- Septa not fused
- 2 cycles septa
- Septa not exsert
- Septal margin not smooth
- Paliform structures absent
Many thanks to Steve Ruddy of Coral Reef Ecosystems (www.coralreefecosystems.com) for his assistance in preparation of this article.
MACNA! Atlanta 2008.www.masna.org
References
- Baker, A., 2003. Flexibility and specificity in coral/algal symbiosis: Diversity, ecology and biogeography of Symbiodinium. Annu. Rev. Ecol. Syst., 34:661-689.
- Battad, J., P. Wilmann, S. Olsen, E. Byres, S. Smith, S. Dove, K. Turcic, R. Devenish, J. Rossjohn and M. Prescott, 2007. A structural basis for the pH-dependent increase in fluorescence efficiency of chromoproteins. J. Mol. Biol.
- Chen, C., Y-W Yang, N. Wei, W-S Tsai and L-S Fang, 2005. Symbiont diversity in scleractinian corals from tropical reefs and sub-tropical non-reef communities in Taiwan. Coral Reefs, 24(1): 11-22.
- Iglesias-Prieto, R., V. Beltran, T. LaJeunesse, H. Reyes-Bonilla and P. Thome, 2004. Different algal symbionts explain the vertical distribution of dominant reef corals in the eastern Pacific. Proc. R. Soc. Lond. B, 271:1757-1763.
- LaJeunesse, T., W. Loh, R. van Woesik, O. Hoegh-Guldberg, G. Schmidt and W. Fitt, 2003. Low symbiont diversity in southern Great Barrier Reef corals, relative to those of the Caribbean. Limnol. Oceanogr., 48(5):2046-2054.
- LaJeunesse, T., R. Bhagooli, M. Hidaka, L. deVantier, T. Done, G. Schmidt, W. Fitt and O. Hoegh-Guldberg, 2004. Closely related Symbiodinium spp. differ in relative dominance in coral reef host communities across environmental, latitudinal and biogeographic gradients. Mar. Ecol. Prog. Ser., 284:147-161.2004.
- LaJeunesse, T., D. Thornhill, E. Cox, F. Stanton, W. Fitt and G. Schmidt, 2004. High diversity and host specificity observed among symbiotic dinoflagellates in reef coral communities from Hawaii. Coral Reefs, 23:596-603.
- Magalon, H., J.-F. Flot and E. Baudry, 2007. Molecular identification of symbiotic dinoflagellates in Pacific corals in the genus Pocillopora. Coral reefs, 26:551-558.
- Mieog, J., M. van Oppen, N. Cantin, W. Stam and J. Olsen, 2007. Real-time PCR reveals a high incidence of Symbiodinium clade D at low levels in four scleractinian corals across the Great Barrier Reef: implications for symbiont shuffling. Coral Reefs, 26:449-457
- Riddle, D., 2006. Temperature and the reef aquarium. Advanced Aquarist Online. http://www.advancedaquarist.com/2006/2/aafeature2/
- Riddle, D., 2007. Product Review: Water motion devices: Sea Flo’s Maxi-Jet modification kits. http://www.advancedaquarist.com/2007/12/review1
- Salih, A., G. Cox, R. Syymczak, S. Coles, A. Baird, A. Dunstan, G. Cocco, J. Mills and A. Larkum, 2006. The role of host-based color and fluorescent pigments in photoprotection and in reducing bleaching stress in corals. Proc. 10th Coral Reef Symp., Okinawa, Japan.
- Ulstrap, K., P. Ralph, A. Larkum and M. Kuhl, 2006. Intra-colony variability in light acclimation of zooxanthellae in coral tissues of Pocillopora damicornis. Mar. Biol., 149:1325-1335.
- Van Oppen, M., F. Palstra, A. Piquet and D. Miller, 2001. Patterns of coral-dinoflagellate associations in Acropora: Significance of local availability and physiology of Symbiodinium strains and host-symbiont selectivity. Proc. R. Soc. Lond B., 268: 1759-1767.
- Van Oppen, M., 2004. Mode of zooxanthella transmission does not affect zooxanthella diversity in Acroporid corals. Mar. Biol., 144:1-7.
- Van Oppen, M., A. Mahiny, and T. Done, 2005. Geographical distribution of zooxanthella types of three coral species of the Great Barrier Reef sampled after the 2002 bleaching events. Coral Reefs, in press.
- Veron, J., 2000. Corals of the World. Australian Institute of Marine Science, Townsville.
- Veron, J., 2002. Coral ID: An electronic key to the zooxanthellate scleractinian corals of the world. (software). Australian Institute of Marine Science and CCR Qld Pty Ltd. University of Queensland, Australia.
- Warner, M., T. LaJeunesse, J. Robinson and R. Thur, 2006. The ecological distribution and comparative photobiology of symbiotic dinoflagellates from reef corals in Belize: Potential implications for coral bleaching. Limnol. Oceanogr., 51(4):1887-1897.




0 Comments