For the past months we’ve discussed culturing micro-foods to satisfy the three steps in the food chain required to feed our potential fish fry. To recap the past columns, we have grown greenwater (phytoplanktons) to feed to our rotifer cultures. We’ve covered the use of rotifers as an initial food source for fry, and then described the culture of ciliates as helper food items for smaller fish larvae. In this month’s column we’ll discuss the use of Artemia (brine Shrimp) nauplii as a food source for larger fry.
Introduction
Among the live diets used in the aquaculture of fish, brine shrimp (Artemia) nauplii are the most widely used food item, mainly due to its convenience and availability. [Fig 1] Unquestionably, the ability of this small branchiopod crustacean to form dormant embryos, called ‘cysts’ (brine shrimp eggs), helps make this food source convenient, suitable, and an excellent larval food source for aquaculture. (Dhont, 1993) These cysts are available in large quantities year round, along the shorelines of hypersaline lakes. After harvesting and processing, Artemia cysts have remarkable shelf life and can be stored in containers for years, to be opened and utilized as a “ready-made live food source.” Reawakening the cysts with overnight hydration in saltwater releases wiggling nauplii that are usable directly as a live food to the larvae of a wide variety of marine ornamentals. The ease and simplicity of hatching brine shrimp nauplii makes them one of the most convenient, least labor-intensive live foods available for aquaculture. Artemia are nonspecific feeders and will ingest a wide variety of foods, which makes them even more useful to aquaculturists. Additionally nutritional enrichment of Artemia can enhance levels of important marine-based highly unsaturated fatty acids (HUFA) to equal many copepod species. Furthermore, the ability of Artemia to feed on floating particles allows the bioencapsulation of specific agents tailored to suit the predators’ requirements. [Fig 2] Use of species-specific bio-encapsulation has had a major impact on improved larval survival, growth, success of metamorphosis and quality of many species of fish and crustaceans. Bio-encapsulation methods are now being developed for oral delivery of vitamins, chemotherapeutics and vaccines that will further boost the importance of Artemia in the aquaculture world.

Fig 1: The life cycle of Artemia. Gif courtesy of Jason Chaulk. http://www.swgc.mun.ca/biology/envs1000/jchaulk
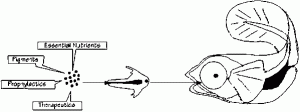
Fig 2: Diagram of the use of Artemia as a delivery vector for transfer of specific components into the cultured larvae diet.

Fig 3 (from top to bottom): Artemia cyst as (a)dispensed from a can of store bought brine shrimp. (b) photomicrograph of cysts, (c) cysts bursting “hatching”
Artemia Life Cycle
The Artemia life cycle begins by the hatching of dormant cysts. These cysts are metabolically inactive embryos that can remain dormant for many years, as long as they are kept dry and oxygen free. When the cysts are placed back into salt water they re-hydrate and resume their development. [Fig 3] After hydration for 15 to 20 hours at 25 oC (77 oF) the cyst bursts and the embryo leaves the shell. While the embryo hangs underneath the empty shell (umbrella stage) the development of the nauplius is completed. Within a short period of time the hatching membrane is ruptured (hatching) and the free-swimming nauplius is born. In the first larval stage, the nauplii (first larval stage -instar I; 400 to 500 µm in length) are a brownish-orange color because their yolk reserves. Newly hatched Artemia do not feed because their mouth and anus are not fully developed. Approximately 12 hours after hatching, the animals molt into the 2nd larval stage (instar II). Small food particles ranging in size from 1 to 50 µm are filtered into the digestive tract. During the next eight days, the nauplii grow and progress through 15 molts before reaching adulthood. Adult Artemia average about 8mm long, but can reach lengths up to 20mm in optimal environments. An adult brine shrimp is approximately 20 times longer and 500 fold larger in biomass than a nauplius. Male brine shrimp possess a paired penis in the posterior part of their trunk, and adult female Artemia can easily be recognized by the brood pouch [Fig 5].
In low salinity and optimal food levels, fertilized females usually produce free-swimming nauplii at a rate of up to 75 nauplii per day (ovoviviparous reproduction). Each female shrimp can produce 10-11 broods over an average life span of 50 days. But under ideal conditions, an adult Artemia can live as long as three months and produce up to 300 nauplii or cysts every 4 days. Under deteriorating environmental conditions, such as high salinity, chronic food shortages and/or cyclic oxygen stress, cyst production is induced. The embryos develop up to the gastrula stage, and are then surrounded by a thick shell. [Fig 6] Formation of this shell initiates a state of metabolic dormancy (diapause), and the cysts are released by the female (oviparous reproduction) where they float to the shoreline and dehydrate. Female Artemia can switch from one mode of reproduction to the other.
Adult Artemia can tolerate brief exposures to temperatures as extreme as -18 to 40oC 104oF. There are differences in optimal temperature for cyst hatching and adult grow-out between strains. The optimal temperature for the San Francisco bay strain is 22oC as compared to 30oC for Great Salt lake Artemia (Bossuyt, 1980). Brine shrimp prefer a salinity of 30-35 (1.022-1.026 specific gravity) and can live in fresh water for about 5 hours before they die.
Feeding
Artemia is a non-selective filter feeder of organic detritus, microscopic algae, and bacteria. Food is not directly consumed, but rather transferred to the mouth in a packaged form. The gap between Artemia legs widens as the legs sweep forward. Water is drawn into this space from below, and small filtering hairs collect food particles from the incoming water stream. Once the appendages sweep backward, water is forced out of this space and the food remains in a groove at the base of the legs. This groove has secretory glands that create adhesive mucus which clumps food particles into balls. Once the food balls are formed, micro hairs move the food packages toward the mouth. The optimal size for food is <50microns for nauplii and < 60 microns for adult brine shrimp.

Fig 6 (top) Close-up of female Artemia uterus showing oviparous formation of cysts. (middle and bottom) two images of male Artemia demonstrating head and body structures. Photos courtesy of Jason Chaulk
Cyst Hatching Requirements
Although hatching small quantities of Artemia cysts is basically very simple, several parameters need to be taken into consideration for a successful hatching:
- hatching container
- aeration
- temperature
- salinity
- pH
- cyst density
- illumination
The best hatching results are achieved in conical bottom containers, aerated from the center bottom. (Lavens, 1990) Cylindrical or square-bottomed tanks will have dead spots in which Artemia cysts and nauplii accumulate. Transparent hatching containers (like 2L soda bottles) permit visual inspection of the hatching mix. This is especially useful when one is harvesting the brine shrimp.
The intensity of aeration should be sufficient to maintain high oxygen levels, because increased hatching has been reported with increasing oxygen level. (Tackaert, 991) Excessive foaming must be prevented, as hydrating cysts will be carried out of the container on the bubbles. As for optimal temperature, optimal hatching occurs in the range of 25-28°C; below 25°C cysts hatch more slowly and above 33°C the cyst metabolism is stopped. Reports from commercial hatcheries suggest that strong illumination (approximately 2000 Lux at the water surface) is essential for maximal hatching, and that this lighting is essential during the first hours after complete hydration, in order to initiate embryonic development (Vanhaecke, 1981). Good circulation is essential to keep the cysts in suspension. The optimal conditions for hatching Artemia are: 250C, salinity – (1.030 density), pH range 8-8.5 with heavy continuous aeration.
Enrichment Of Brine Shrimp
As mentioned in the introduction, a critical factor affecting the nutritional value of Artemia as a food source for marine larvae is the content of essential fatty acids. Most marine fish fry are incapable of synthesizing these HUFAs, and therefore must be provided with these fatty acids in their diets. Therefore to ensure adequate HUFAs, we can provide fatty acids as food supplements to Artemia nauplii. Once the nauplii have molted past the second larval stage (about 8 hours after hatching), one can simply include lipid products into the culture media of the brine shrimp nauplii. This technique of bioencapsulation, also called Artemia enrichment (or boosting) and is widely used in fish hatcheries. Other enrichment products have been created to boost specific aspects of the predator nutritional diet, and these products include feeding phytoplanktons, yeast, micro-particulate diets and self-emulsifying concentrates to the nauplii. According to (Lavens, 1987) the highest enrichment levels can be obtained by adding emulsified concentrates, such as Selco to the Artemia’s diet. Selco (also known as Selcon or super Selco) is a self-dispersing complex of selected marine oil sources, vitamins, and carotenoids and upon dilution in seawater disperses into microparticles that are readily ingested by Artemia.
Decapsulated Cysts
The hard egg shell that covers a Artemia cyst can be completely removed by short exposure to a bleach solution. This procedure is called decapsulation and decapsulated cysts offer a number of advantages to the home hobbyist compared to non-decapsulated ones:
- Brine shrimp egg shells are not introduced into the culture tanks. When hatching normal cysts, the separation of nauplii from their shells is not always possible. Unhatched cysts and empty shells can cause deleterious effects in the larval tanks when they are ingested by the fry, often times the egg casing can not be digested and may obstruct the gut.
- Nauplii that are hatched out of decapsulated cysts have a higher energy content and individual weight (30-55% depending on strain) than regular nauplii.
- Decapsulation results in a disinfection of the cysts. A culture infection due to contamination by “dirty” brine shrimp eggs is common in aquaculture facilities, fortunately not so much in home culture.
- Decapsulated cysts can be used immediately as an energy-rich food source for fish and shrimp larvae.
- The illumination requirements for hatching decapsulated cysts are lower.
The procedure to decapsulate Artemia cysts requires an initial hydration of the cysts (as complete removal of the envelope can only be performed when the cysts are spherical), removal of the brown shell in a bleach solution, followed by washing and neutralization of the remaining bleach. These decapsulated cysts can be hatched into nauplii immediately, or dehydrated in concentrated brine solution then stored for later hatching. Lastly, the decapsulated eggs can be used for direct feeding. Decapsulated Artemia cysts can be stored for a few days in the refrigerator without any noted decrease in hatching. Okay enough of the scientific background; let’s get into the important part.
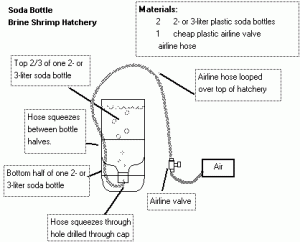
Fig 7: The 2 L soda bottle brine shrimp hatchery. Shown here is a basic layout of what is needed to construct this simple Artemia hatchery. Image courtesy of Matt Lindenfelser (http://www.killi.net/)
Baby Brine Shrimp
Baby brine shrimp are an excellent food source for larger fish fry. Usually after initial feedings on rotifers or copepod larvae, fish fry require a large food type. Additionally, baby brine shrimp make a good substitute macro- plankton for some filter-feeding invertebrates.
Home culture of Brine Shrimp
Hatching brine shrimp is quite simple. A brine shrimp hatchery can be built out of almost anything cone shaped. Household items such as 2L soda bottles make excellent hatcheries. If you prefer to go the high-tech route, transparent 1 gal inverted cone hatcheries are available online. Many local pet stores sell Sally’s’ shrimp hatcheries and these hatcheries utilize a sturdy rubber base in which an inverted 2L soda bottle is attached. Again almost any container will work, however ensure that the container has a conical bottom as this ensures water flow and prevents dead spots. Airline tubing pressed to the very bottom of the container is connected to an air pump, and your hatchery is complete.[Fig 7]
Hatching of brine shrimp consists of adding brine shrimp cysts to seawater. Wilkerson recommends a low-salinity, cost-effective method, using the cheapest aquarium salt you can find. (Wilkerson, 1998) Add 2L of dechlorinated tap water and 2 teaspoons of aquarium salt. Once the salt is dissolved, aerate for 1 hour and then mix 1/4 teaspoon (or less) of brine shrimp cysts. Sufficient aeration is critical for a good hatch rate and to ensure the nauplii remain healthy. Airstones are not recommended; therefore, a rigid airline tube can be used and pressed to the bottom of the hatchery. Once the air pump is started, do not be timid in providing air to the culture. The amount of bubbling can reach a rolling boil. While the cysts are hydrating, that many of the cysts may get stuck on the bubbles that are formed at the container surface and be carried out of the water, so a lower initial aeration rate may be helpful. Ensure that your shrimp hatchery is near a light source for the first day and within 16-20 hrs later you should observe baby brine shrimp. These freshly hatched nauplii are the most nutritious and can be feed directly to your fish fry. The addition of the yolk sac causes the nauplii to appear orange. As an aside, if you are feeding clownfish fry, you must use freshly hatched or no more than 30 hr old nauplii — waiting any later nauplii will be too large for the fry to ingest and unless they have been enriched are now depleted of nutrients. So, if you do wait for 24 hrs, the next day the egg yolk will be depleted and the nauplii are feeding and can be enriched by the addition of a particulate food to their media.

Fig 8: 2L soda bottle hatcheries. Shown are 3 brine shrimp hatcheries and 4 bottles of phytoplankton. The airline tubing have been removed from the hatcheries to collect the nauplii. The phytoplankton is used to enrich the nauplii. Photos courtesy of Ray Jay (http://www.angelfire.com/ab/rayjay/)
Feeding and enriching Artemia
Artemia are non-selective filter feeders and therefore will ingest a wide range of foods. The main criteria for food selection are particle size, digestibility, and nutrient levels. (Dobbeleir, 1980) Possibly the best foods for Artemia are live microalgae such as Nannochloropsis, Tetraselmis, Isochrysis, and Pavlova (see home phytoplankton culture). In fact combinations of live phytoplanktons fed to Artemia cultures have demonstrated superior enrichment characteristics (i.e., increased HUFAs) over feeding single phytoplankton species. (d’Agostino, 1980) [Fig 8] However, not all species of unicellular algae are appropriate for sustaining Artemia growth. For example, Chlorella, and Stichococcus have a thick cell wall that cannot be digested by Artemia. Moreover, Artemia cultures can be enriched by feeding a wide variety of processed foods, which is very convenient to the home fish breeder. Processed foods include yeasts (such as brewers yeast), fish meal, soybean powder, egg yolk, and micronized rice bran. Preserved microalgae such as phytoplankton cryopastes (such as Tahitian blend from www.brineshrimpdirect.com) have also been used with success. One problem with using processed foods is that you have to be careful not to overfeed; processed foods will quickly foul the Artemia culture. The simplest way to measure food levels in the Artemia cultures is by visually assessing the clarity of the water. According to Schumann (website below) this is easily accomplished with a small wooden dowel (at least 30cm long) in which measuring marks (marked off in centimeters) are notched into the dowel; on the end of this dowel attach a white disk with black fields, like a small Secchi disk (http://sebagolakeassc.org/secchi.htm). The depth at which the contrast between the white and black fields on the disc disappears determines the light penetration into the tank. Increasing amounts of food in the culture decreases optical transparency. With a density of 5000 nauplii per liter, the transparency should be 15-20 cm for the first week, and 20-25cm thereafter. For optimal production and enrichment of nauplii, it is best to maintain an optimal food level in the culture media, so frequent or continuous feedings work best. I find feeding of phytoplankton to the Artemia cultures works extremely well and fry fed phytoplankton-enriched nauplii grow quickly. To supplement the phytoplankton I also recommend the use of selcon enrichment to boost HUFA levels. For enrichment, 24-30 hr old nauplii can be transferred into a new culture container, where enrichment emulsion (Selcon) is added at 1 ml/liter and aeration is started. Six hours later, the enriched nauplii are harvested. In my experience there is no need to rinse the nauplii; use them directly.
Harvesting Nauplii
Harvest the nauplii by simply turning off the air, and allow the culture to settle for approximately ten minutes. Hatched, empty shells float to the surface, and unhatched cysts will sink to the bottom. The newly hatched nauplii will concentrate just above the unhatched cysts on the bottom. Since the newly hatched nauplii are attracted to light, shining a flashlight at the center of the bottle will concentrate the nauplii where it is easy to siphon them off, or in a commercial hatching tank, one can drain the cysts off the bottom by using the valve, then drain the nauplii onto another container. An alternative method to collect nauplii is to utilize a brine shrimp net (which is a small mesh material) after decanting the floating hatched eggs off the top. An inexpensive siphon can be constructed out of rigid air (1/8″) tubing with 2-3 ft of flex tubing attached. Then just siphon the wriggling shrimp off into a brine shrimp net, and add to your fish fry growout tank.
Sources Of Brine Shrimp Eggs
Eggs can be bought in most pet shops or by mail order (see online sources below). Eggs bought in bulk (such as 1 lb cans) will be less expensive than the tiny tubes sold in stores and will often be fresher and in better condition. The cans may be held in the freezer, with 2-3 weeks worth of supply held in a small, sealed jar.
Fig 9: 16oz can of brine shrimp cyst and home 2L hatchery. This is a common setup for the home culturist. This can will provide brine shrimp nauplii for 6months to 1yr depending on the amount of nauplii needed. In between uses this can should be refrigerated to preserve viability. Photo courtesy of James Wiseman
Maintenance Of The Brine Shrimp Culture
Most people who hatch brine shrimp do so in a low volume batch. Once the nauplii are hatched, the culture is fed, and utilized for 2 or 3 days. After 3 days, the culture is often depleted of nauplii and a new culture, that was started the day before replaces the previous one. Some people desire a continuous culture, and this presents a few new problems. In smaller containers, water quality can deteriorate rapidly, especially as biomass and feeding increase. Furthermore, excess foods not eaten by the Artemia will decompose in the culture medium by bacteria, thereby deteriorating water quality due to a gradual build up of toxic substances such as ammonia and nitrite. Try to prevent overfeeding by observing the amount of food added during each feeding and wait until the culture begins to clear before you add more food.
Once a sizable population of nauplii is generated, it is often difficult to balance the need to feed the culture, and the resulting decrease in water quality from the pollution that feeding causes, especially when non-living feeds are used. Performing a 50% water change weekly will help overcome this problem. Additionally, siphoning and cleaning the bottom of the culture vessel every few days will remove accumulated detritus and prevent further water quality issues. If the goal is to have long-term culture then one should consider adding a small sponge filter or other nitrifying filter to the culture.
Troubleshooting an Artemia Culture
The two problems I have repeatedly encountered when culturing Artemia, are slow growth and the culture crashing. Slow growth is usually attributed to environmental issues such as low temperatures, an improper pH, too low or too high salinity, and insufficient food or poor food quality. There are several possible causes for a Artemia culture crash. The easiest problem to resolve is low dissolved oxygen. This is remedied by increasing the amount of air and amount of water motion thru the culture. Secondly, the health status of Artemia can be visually assessed by observing their swimming motion. Healthy cultures react to photo stimuli, and rapidly gather at the light source. Slow, dispersed swimming indicates that the animals are in poor condition. Health can also be assessed by visually inspecting the Artemia under a microscope. Well-fed Artemia have a completely filled gut and release compact fecal pellets. They should also have clean swimming appendages and a clean mouth region suggesting that the animal is in good health. Underfed animals have an empty or barely filled gut and tend to release loose fecal pellets. If their mouth parts or appendages are covered with food particles, this suggests the animal is feeding poorly. Another reason for crashing cultures is a bacterial or protozoan contamination of the culture. This can be avoided by decapsulating the cysts before hatching.
Conclusion
Brine shrimp nauplii are an easy and convenient food source for larger fish fry. The ability to nutritionally enrich Artemia nauplii provides us with a delivery platform to specifically target potential predators’ nutritional requirements and met these needs. Feeding and providing proper nutrition to fish larvae is the greatest hurdle faced by all potential marine fish breeders. With a bit of closet space, spare tanks, extra aquarium supplies, and the information provided in the past Breeder’s Net columns, virtually any marine hobbyist should be able to raise enough live food to consider breeding marine fish at home. Over the next few months we’ll begin to produce columns on the breeding requirement of specific fish species suitable for home culture, plus information on additional micro foods.
How to Decapsulate Brine Shrimp eggs
Hydration Step
- Hydrate cysts by placing them for 1 h in water (< 100 gm/l), with aeration, at 25°C.
Decapsulation Step
- Collect cysts on a 125 µm mesh sieve, rinse, and transfer to the hypochlorite solution.
- The hypochlorite solution can be made up (in advance) of either liquid bleach NaOCl (fresh product; activity normally =11-13% w/w) or bleaching powder Ca(OCl)2 (activity normally ± 70%) in the following proportions:
- 0.5 g active hypochlorite product (activity normally labeled on the package, otherwise to be determined by titration) per g of cysts; for procedure see further;
- an alkaline product to keep the pH>10; per g of cysts use:
- 0.15 g technical grade NaOH when using liquid bleach;
- either 0.67 NaCO3 or 0.4 g CaO for bleaching powder; dissolve the bleaching powder before adding the alkaline product; use only the supernatants of this solution;
- seawater to make up the final solution to 14 ml per g of cysts.
- Cool the solution to 15-20°C (i.e. by placing the decapsulation container in a bath filled with ice water). Add the hydrated cysts and keep them in suspension (i.e. with an aeration tube) for 5-15 min. Check the temperature regularly, since the reaction is exothermic; never exceed 40°C (if needed add ice to decapsulation solution). You can assess how well the process is working by checking for decapsulation under a jewelers eyepiece or microscope.
Washing Step
- When cysts turn grey (with powder bleach) or orange (with liquid bleach), or when microscopic examination shows almost complete dissolution of the cyst shell (= after 3-15 min.), cysts should be removed from the decapsulation suspension and rinsed with water on a 125 µm screen until no chlorine smell is detected. It is crucial not to leave the embryos in the decapsulation solution longer than strictly necessary, since this will affect their viability.
Deactivation Step
- Deactivate all traces of hypochlorite by dipping the cysts (< 1 min.) either in 0.1 N HCl or in a 0.1% Na2S2O3 solution, then rinse again with water. Hypochlorite residues can be detected by putting some decapsulated cysts in a small amount of starch-iodine indicator (= starch, KI, H2SO4 and water). When the reagent turns blue, washing and deactivation has to be continued. Alternatively, a DPD chlorine test method may be used.
Use
- Incubate the cysts for hatching, or store in the refrigerator (0-4°C) for a few days before hatching incubation. For long term storage cysts need to be dehydrated in saturated brine solution (1 g of dry cysts per 10 ml of brine of 300 g NaCl.l-1). The brine has to be renewed after 24h.
References
- Bossuyt, E.; Sorgeloos, P. (1980). Technological aspects of the batch culturing of Artemia in high densities. The Brine Shrimp Artemia, Ecology, Culturing, Use in Aquaculture 3: 133-152.
- D’Agostino, A.S. 1980. The vital requirements of Artemia: physiology and nutrition. In: The Brine Shrimp Artemia, Vol. 2, Physiology, Biodiversity, Molecular & Biology. G. Personne, P. Sorgeloos, O. Roels and E. Jaspers (Eds). Universa Press, Wetteren, Belgium, pp 55-82.
- Dobbeleir, J.; Adam, N.; Bossuyt, E.; Bruggeman, E.; Sorgeloos, P. (1980). New aspects of the use of inert diets for high density culturing of brine shrimp. The Brine Shrimp Artemia, Ecology, Culturing, Use in Aquaculture 3: 165-174
- Dhont J., Lavens, P. and Sorgeloos, P. 1993. Preparation and use of Artemia as food for shrimp and prawn larvae. In: CRC Handbook of Mariculture. 2nd Edition. Vol 1: Crustacean Culture. J.V. Mc Vey (Ed). CRC Press, Inc., Boca Raton, Florida, USA, pp 61-93.
- Lavens P., and Sorgeloos, P. 1991. Chapter XIII : Production of Artemia in culture tanks. In: Artemia Biology. R.A. Browne, P. Sorgeloos and C.N.A. Trotman (Eds). CRC Press, Inc. Boca Raton, Florida, USA, pp 317-350.
- Lavens P., and Sorgeloos, P. 1987. Design, operation, and potential of a culture system for the continuous production of Artemia nauplii. In: Artemia Research and its Applications. Vol. 3. Ecology, Culturing, Use in Aquaculture P. Sorgeloos, D. Bengtson, W. Decleir and E. Jaspers (Eds) Universa Press, Wetteren, Belgium: 339-345.
- Tackaert, W. and Sorgeloos, P. 1991. Semi-intensive culturing in fertilized ponds: 287-315. In: Artemia Biology. Browne, R.A., Sorgeloos, P. and C.N.A. Trotman (Eds), CRC Press, Inc., Boca Raton, Florida, USA, 374 p.
- Vanhaecke, P.; Cooreman, A.; Sorgeloos, P. (1981). International study on Artemia : 15. Effect of light intensity on hatching rate of Artemia cysts from different geographical origin. Mar. Ecol. Prog. Ser. 5: 111-114.
- Wilkerson, J., Clownfishes, Microcosm Limited; ISBN: 1890087041; June 1998
Online Reading
- Brine Shrimp Directs website: http://www.brineshrimpdirect.com/brineshirmparticles1.html
- Kai Schumann Brine shrimp page: http://www.angelfire.com/wa/AquariaWeb/_Artemia_.html
- Matt Lindenfelser-Do it yourself Brine Shrimp Hatchery: http://www.killi.net/foodarts/brine.html
- Brine shrimp info page: http://www.intellweb.com/gcka/fishroomfoods.htm
- Sach’s Aquaculture site for Brine shrimp foods: http://www.aquaculturestore.com/info/recipes.html
- Raising Adult Brine Shrimp: http://www.trakkerinc.com/tropfish/articles/tf06/tf060903.htm









Hi Frank,
Do you have any contact details for Jason Chaulk please? I am interested in using one of his images in a biology teaching unit.
Any help you can give in helping me track him down would be much appreciated.
Kind regards,
Will