“Is it possible to have too much light over my reef aquarium?” is a simple – and legitimate – question. Opinions widely vary on just how much light is enough (or too much). While excessive light in a natural environment is known to cause problems for plants, algae and zooxanthellae, some debate remains if it is possible to provide too much light – especially for small-polyped stony corals – in an artificial setting. This brief article will present results of an experiment in which a coral was exposed to high intensity artificial light within a setting likely replicated by many reef aquaria. It will also discuss concepts well-established within the world of botanical research -those of the intimate and intricate world of photosynthesis, and provide insight of the dynamics of photosynthesis when a coral is exposed to sudden, intense artificial light.
Introduction
Advances in instrumentation over the last two decades have made possible non- intrusive means of examining the kinetics of photosynthesis (however, see limitations imposed by insufficient water motion in the Discussion section). A notable advance has been that of modulated pulse fluorometry. One of these instruments, a pulsed amplitude modulation (PAM) fluorometer, examines chlorophyll fluorescence and is able to determine how energy is used (and not used) by photochemical reactions. In essence, a PAM chlorophyll fluorometer is a ‘photosynthesis meter,’ and allows one to gain insights of photochemical and non-photochemical reactions.
Chlorophylls are abundant photopigments and, along with accessory or antennae pigments, harvest light energy. By-products of molecular oxygen and organic carbon are ultimately produced through the process known as photosynthesis.
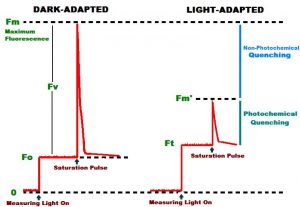
If a form of chlorophyll – chlorophyll a – is exposed to strong light, it will absorb a portion of the light’s energy and use it in photosynthesis. Chlorophyll a will also absorb and emit some of this light’s energy at a lower energy level in a phenomenon known as fluorescence. Fluorescent emissions of chlorophyll a are generally considered red, and are known to range from 660 nm to 760 nm. Plants, marine algae, and corals with healthy symbiotic dinoflagellates will fluoresce when exposed to relatively high amounts of visible light. Chlorophyll fluorescence is proportional (to a point) to the amount of photosynthetically active radiation. If no light energy is available for, say, 20 minutes, chlorophyll fluorescence is, for all intents and purposes, at zero, so a very weak amount of light (< 1 µmolm2sec) is applied by a PAM meter to cause chlorophyll a to weakly fluoresce. This is measured and reported as Minimum Fluorescence (Fo). If a brief pulse of intense, photosynthetically saturating light is applied to a dark-adapted sample, the fluorescence will rise to a maximum level. This is called Maximum Fluorescence (Fm – See Figure 1). It is also possible to estimate ‘variable’ fluorescence (Fv) simply by subtracting Fo from Fm. Fluorescence of an illuminated sample during a saturating pulse of light when all PS II reaction centers are saturated with light (‘closed’), is called Maximum Fluorescence (Fm’ – the prime symbol indicates an illuminated sample). Fm’ is generally less than Fm. If one were to subtract Fm’ from Fm, the difference is due to ‘non-photochemical reactions’ (denoted as qN or NPQ, depending upon circumstances). Non- photochemical reactions compete with photochemical reactions in ‘quenching’ (suppressing) maximum fluorescence. Thus, results from measurements of ‘minimum,’ ‘variable’ and ‘maximum’ fluorescence can be manipulated mathematically to determine how light energy is used and/or dissipated.
Note: This fluorometer uses a ‘red’ light-emitting diode (LED) as ‘actinic’ light. This light energy is absorbed by PS II’s reaction center, containing Pigment 680 (P-680). Since P-680 (a specialized form of chlorophyll a) absorbs energy collected by chlorophylls a, c2 and accessory pigments such as peridinin, the red excitation bandwidth is appropriate for use with zooxanthellae. A light filter (cutoff λ <680 nm) prevents the PAM’s internal photo-amplifier from confusing excitation wavelengths for those of chlorophyll fluorescence. There are some advantages (and disadvantages) of using a ‘blue’ LED as an actinic source, however, these do not dismiss the results gathered while using red wavelengths as an excitation source.
These formulae are used to determine photochemical efficiencies:
- Photochemical Quenching (qP)
- (Fm’-Ft)/ (Fm’-Fo). qP is the energy absorbed by PS II.
- Non-photochemical Quenching (qN)
- (Fm-Fm’)/ (Fm-Fo). qN is generally associated with non-photochemical activity, such as dissipation of absorbed energy as heat, or as a thylakoid-energizing prelude to photosynthesis.
- Non-photochemical Quenching (NPQ)
- (Fm-Fm’)/Fm’. NPQ is particularly associated with energy dissipation as non-radiant heat through the ‘xanthophyll cycle.’
- Yield of Photochemical Energy Conversion
- (Fm’-Ft)/Fm’ = ΔF/Fm’
Light-harvesting Structures Of Zooxanthellae
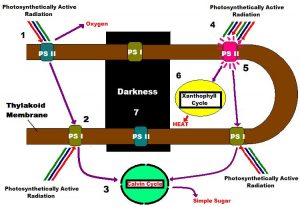
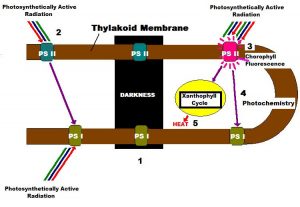
Light-harvesting photopigments within zooxanthellae are found in thylakoid membranes contained within structures called chloroplasts (See Figure 2). It is thought that one Photosystem I and one Photosystem II are arranged within a few microns of each other on the thylakoid, so that they may act effectively transfer energy. These photosystems combined are known as a Photosynthetic Unit, or PSU (Kirk, 2000).
- A moderate amount of light (or PAR – photosynthetically active radiation) falls upon one of the many thylakoid membranes (brown line) containing a Photosynthetic Unit (consisting of one Photosystem II and one Photosystem I) within a healthy zoxanthella. Photosystem II (PS II) photopigments absorb PAR and transfer its energy to a ‘Reaction Center’ within PS II. Note that some variable chlorophyll a fluorescence occurs even at moderate light intensity, as Reaction Centers absorb light and begin to ‘close.’ One oxygen molecule is created for every two water molecules split.
- Light energy collected by PS II is transferred to PS I (specifically the PS I Reaction Center). PS I photopigments also collect PAR transfers energy to:
- The Calvin Cycle, where inorganic carbon is converted to simple sugar.
- Under conditions of high PAR intensity, ‘safety valves’ for excess light energy come into play. PS II reaction centers absorb as much energy as they can (photosynthesis is said to be ‘saturated’ when all reaction centers are ‘closed’), and two safeties dump excess energy:
- Chlorophyll fluorescence, which depends upon the number of ‘closed’ reaction centers and:
- Transfer of energy to the ‘_Xanthophyll Cycle_,’ where energy absorbed by the antennae pigments is dissipated as non-radiant heat, and involves the reversible, light-mediated conversion of diadinoxanthin to diatoxanthin within zooxanthellae of corals.
- In darkness, PS II chlorophyll fluorescence is at a minimum, and no photochemistry occurs. However, the Xanthophyll Cycle continues with conversion of diatoxanthin back to diadinoxanthin.
- In darkness, minimal fluorescence of PS II chlorophyll is found. In other words, no energy is available to the photosystem, and it is fully ‘open’ (oxidized) and ready to absorb light energy. A small amount of light energy is applied to the sample, and this induces chlorophyll fluorescence, known as Minimal Fluorescence Yield following dark adaptation, and noted as Fo. If a saturating pulse of light is applied to the dark-adapted sample, the Reaction Centers are ‘closed’ (reduced) and chlorophyll fluorescence will be at its highest value. Maximum Fluorescence of a dark-adapted sample is called Fm.
- When illuminated with non-saturating light intensity, the chlorophyll molecules of PS II begin to fluorescence as Reaction Centers are reduced (closed). This fluorescence is known as Variable Fluorescence (Fv, which is equal to Fm – Fo – or Fm’ – Fo’, see below).
- The Maximum Fluorescence of the illuminated sample is known as Fm’.
- Under conditions of proper illumination (and other environmental conditions), the Electron Transport Rate (ETR) will continue between Photosystems I and II. Photosynthesis is said to be ‘saturated’ when the amount of PAR available to the photosystems meets or exceeds the maximum rate absorbed by the photosystem, and increasing the amount of light will not increase the rate of photosynthesis. A method of energy dissipation – other than photochemical quenching (i.e., the absorption of energy used in photochemistry and called qP) – must then be used, and this is known as:
- ‘Non-photochemical quenching’ or NPQ. NPQ involves the ‘Xanthophyll Cycle’ where protective pigments dissipate excess harvested photons as non- radiant heat.
Procedure
A small “Rice Coral” ( Montipora patula, see Figure 5) colony was selected from one of NELHA’s (Natural Energy Laboratory of Hawaii) outdoor 75-gallon aquaria, which have a constant flow-through of seawater pumped from a depth of 13 m. Even though shade cloth is used to attenuate natural sunlight, all animals housed within this tank receive a maximum light intensity of 800 μmol∗m2∗sec.
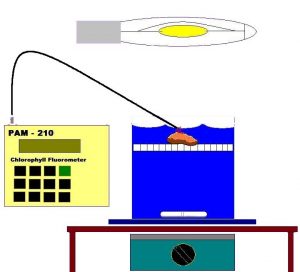
This coral colony was transported within a 19-liter plastic bucket, filled with seawater, to a darkened and air-conditioned laboratory. The coral was transferred to a round 4 liter chamber with a false bottom of plastic ‘egg crate’ material. The container was also filled with natural seawater. (See Figure 6).
A magnetic stirrer and large stir bar provided relatively constant water motion within the chamber. After a 30-minute ‘dark-adaptation’ period (to allow PS II Reaction Centers to ‘open’), the PAM 210 Chlorophyll Fluorometer (Heinz Walz GmbH, Effeltrich, Germany), equipped with a submersible fiber optic cord (approximately 1.5 mm diameter), assessed zooxanthellae Fo and Fm fluorescence, after correction for instrument signal noise (Zero Offset). The tip of the probe was positioned to monitor the tissue between the polyps – the coenosarc. A 400-watt, 6500K metal halide lamp* was then used to illuminate the coral sample and, when fully ‘warmed’, delivered a maximum of 645 μmol∗m2∗sec at the coral’s surface. The lamp was approximately 20 cm from the water’s surface. Lexan Solar™ acrylic material, placed atop the chamber, attenuated ultraviolet radiation (<390 nm) to just a few microwatts/cm, and also absorbed heat energy generated by the lamp. A small household fan (combined with the room’s air conditioning) kept the container’s water temperature fairly constant, though a slight rise in temperature from 26° to 27° C was noted over the course of the experiment (approximately 60 minutes). This temperature is not thought to harm at least some zooxanthellae and coral species (Jones et. al., 1998), and is certainly below the upper thermal limits of 32 – 36º C reported by Hoegh-Guldberg (1999) and Fitt and Warner (1995).
The PAM meter’s Saturation Light was set at the maximum setting and provided pulses of light through the fiber optic cable amounting to 791μmol∗m2∗sec (approximately the same as the maximum intensity normally experienced by this coral at noon). These parameters were monitored during the experiment: Minimal Fluorescence (dark-adapted, or Fo), Maximum Fluorescence (dark-adapted, or Fm), Variable Fluorescence (Fv), Fluorescence at a given time (Ft), Maximum Fluorescence (Fm’), qP (Photochemical Quenching), qN (Non-photochemical Quenching) and NPQ (Non-photochemical quenching).
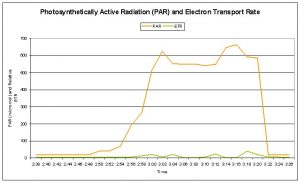
Figure 7: The 400-watt lamp was turned on at 2:48, and turned off at 3:20, for an exposure time of 32 minutes. Maximum PAR reached 662 μmol∗m2∗sec.
*I am reluctant to state the brand name of this lamp, but will say it is a staple in the aquarium trade, and has been for years. The purpose of the experiment was to examine the response of zooxanthellae to sudden, intense artificial lighting, and it is believed (but not proven) the results would have been the same with any other lamp.
Results
Figures 7 and 8 demonstrate the dynamics of photosynthesis within zooxanthellae of the Montipora patula specimen. Photosynthetic Yield and Photochemical Quenching decreased with increasing radiation, while Non- photochemical Quenching increased.
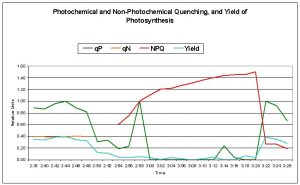
Figure 8: Photochemical Quenching (qP) collapsed after approx. 12 minutes exposure to the increasing amount of visible light (but filtered for ultraviolet radiation). Maximum qP was 0.398 just before exposure, and fell to zero. Since photochemistry was not dissipating absorbed light energy, Non- photochemical Quenching (NPQ) dumped it as non-radiant heat.
Discussion
Strong illumination apparently caused drastic changes within the photochemical reactions, and seems to indicate a symbiotic relationship in distress. This suggests that photoinhibition can indeed occur at relatively low light intensity – we see the possibility of sharply reduced photosynthetic rates at only 260 μmol∗m2∗sec. Light intensity of this level is certainly within the potential of efficient lighting systems, including standard, VHO and PC fluorescent lamps, and metal halide bulbs of singular (or combined) wattages of about 200 and upwards. Significant non-photochemical quenching (NPQ, exceeding 0.5) is seem at lower light intensity – that of ~100 μmol∗m2∗sec.
Montipora patula colonies are generally considered shallow-water corals, most often found high on reef slopes or in shallow bays that afford protection from strong wave action (Gulko, 1998). This colony was no exception, and was legally collected at a depth of about 8 m off the west shore of the big island of Hawaii. This particular colony was fully exposed to sunlight at depth (at a maximum PAR level estimated to be approximately 1,000 μmol∗m2∗sec at noon on a cloudless day).
This Montipora patula specimen has been maintained for months in captivity, where natural sunlight delivers a maximum of about 800 μmol∗m2∗sec for several hours daily. This coral has likely photoacclimated to high intensity light as much as possible, yet signs of dynamic photoinhibition are seen at relatively low light intensity, with a collapse of photochemical quenching (qP) at ~32% of the normal maximum light intensity in captivity.
Non-photochemical quenching (qN), was measured at about 0.4 during the first few minutes of the experiment, when the metal halide lamp was ‘off’ and the only available light energy originated from the low output from the LEDs of the PAM fluorometer. qN, at this low value, is associated with ‘thylakoid membrane energization.’ When qN values exceed 0.5, a different measurement of non-photochemical quenching is used – NPQ – which is sensitive to that portion of non-photochemical quenching which demonstrates dissipation of excessive PAR as non-radiant heat (Schreiber, 1997). Thus, qN is reported during the first few minutes of the experiment, followed by neither qN or NPQ being reported during the transitional phase just after the lamp was turned on, and, finally, NPQ as it exceeds a value of 0.5, and is therefore a convenient indicator of ‘too much light.’ Certainly, the existence of NPQ within this coral colony presents a strong case for the presence of dynamic photoinhibition by xanthophylls.
One should include a ‘lag factor’ of several minutes when viewing the results. Since some photoreactions occur relatively slowly, Schreiber (1997) advises measurements of ‘photochemical quenching’ and ‘non-photochemical quenching’ should take place only after a photosynthetic sample has been illuminated for about 2 minutes to allow these ‘slow’ reactions of photosynthesis to occur.
There is a possibility that the PAM meter’s fiber optic cord, in close proximity to the coral (distance of 2 mm), may have created a thickened boundary layer around the examined area, and potentially resulted in limitation in diffusion of nutrients (possibly nitrogen, iron, phosphorus, etc. – See Gorbunov et al., 2000; Atkinson et. al., 1994; Atkinson and Bilger, 1992). Ralph et al., 2002, recommend use of small fiber optic cables in order to differentiate between rates of photosynthesis within coenosarc and polyp tissues as well as to minimize the impact of the probe’s presence upon the results. With that said, the magnetic stirrer was adjusted to provide flow velocities of 15 cm/sec within the chamber (judged visually), and it is felt that adequate water motion to create turbulence across the coral colony was provided. There is an important point here – water motion within an aquarium takes on added importance if a small obstruction could indeed cause this sort of
reaction within zooxanthellae. The speed of the current within the chamber (15 cm/sec) is close to maximum velocities measured on sheltered Hawaiian reefs during calm weather (Riddle, unpublished). Having used a digital water velocity meter to measure water motion in hundreds of aquaria, I can say with some justification that most cannot match, or even approach, ‘natural’ water movement.
One should recall that this experiment was conducted with nutrient-poor natural seawater. Artificial seawater mixes are generally enriched with micro- nutrients in respect to oceanic waters (Atkinson and Bingman, 1999), and matured aquaria water tends to contain elevated concentrations of micro- and macro-nutrients (Atkinson et al., 1995). Interestingly, nutrient deficiency (specifically that of nitrogen) of symbiotic zooxanthellae is thought to cause the relatively low yield of photosynthesis (an Fv/Fm of only 0.39, as opposed to 0.50 to 0.75 for seagrasses, and 0.65 for many plankton species (Falkowski and Kolber, 1995; Gorbunov et. al., 2000), and about 0.80 for terrestrial green plants – personal observations). Photosynthetic yields ranged from 0.62 to 0.66 in zooxanthellae isolated from corals and grown in nutrient-rich conditions (Kolbert et. al., 1988). Compare this information with that of Bongiorni et.al, 2003, that reports relatively high growth rates of stony
corals exposed to elevated nutrient levels generated by a nearby commercial fish farm. The notion that slight fertilization of symbiotic zooxanthellae can profoundly affect photosynthetic activity is certainly intriguing.
Photosynthetic Yield returned to ‘normal’ levels only minutes after the 400-watt lamp was extinguished, indicating exposure to the UV-filtered, but saturating, radiation for approximately 1 hour did not cause lasting (chronic) photoinhibition damage to the photosynthetic apparatus (as indicated during the last few minutes of the experiment by the rapid drop in NPQ and concurrent increases of both Yield and Photochemical Quenching).
Calvin Cycle Inhibition is indicated by a noticeable drop in electron transport rate concurrent with strong enhancement of energy dependent non- photochemical quenching (Jones et al., 1998). The results of this experiment indicate a sharp drop in photosynthetic yield and rapid rise in non- photochemical fluorescence quenching. The electron transport rate rose, albeit slightly, only by virtue of very strong illumination combined with extremely low yields. Note that photochemical quenching practically collapsed within minutes after initiation of illumination. It is believed that the inability of the ‘dark reactions’ to absorb energy creates a traffic jam of electrons within PS II, which could create singlet oxygen within the tissues, potentially leading to permanent damage of photopigments and associated structures should these processes continue for a prolonged period.
It is interesting to note that there is a casual relation between photochemical quenching and lower PAR levels during the ‘lighted’ portion of the experiment (Photosynthetic Yield and ETR increased when PAR dropped below 550 μmol∗m2∗sec.). The reason for this is unclear. Down-regulation of photosynthesis during periods of high light intensity should be expected (for instance, see Ralph et. al., 2002 for relative electron transport rates for six coral species).
These results present a case for dynamic photoinhibition within the zooxanthellae of this captive coral, and suggest over-lighting is indeed a possibility in artificial conditions, even if a small-polyped stony coral is photoacclimated to high light intensity.
Many questions are asked. How do nutrient levels and heavy metal concentrations affect photosynthetic yields? Can a balance of increased nutrients (as suggested by Sprung, Delbeek and others) and reduced lighting achieve maximal coral growth rates? If ‘fertilized’ zooxanthellae compete with the calcification process for carbon, how would an increase of alkalinity affect the rate of photosynthesis, and ultimately, coral growth? How would UV-A, UV-B and UV-C from unshielded double-ended metal halide lamps influence photosynthesis? Future projects will include examination of zooxanthellae photosynthetic capacities in nutrient-enriched aquarium water and examination of effects of artificially-produced UV energy.
As a footnote, I will bring the PAM meter to the Boston MACNA if anyone is interested in seeing this device at work, and perhaps we can gather some meaningful data from your coral samples. I’m especially interested in examining fluorescent SPS fragments, but practically no work has been done with LPS or soft corals, and your samples could provide important, new information. Contact me at [email protected] if you’re interested.
Acknowledgements
Mahalo to Sara Peck, University of Hawaii SeaGrant, for her patience and support, Charles Delbeek for the heads up on the Bongiorni et al. reference and to Julian Sprung for thoughtful reflections on aquaria nutrients and reduced lighting.
Glossary
- Fo
- Minimal Fluorescent Yield after dark adaptation.
- Fo’
- Minimal Fluorescent Yield of illuminated sample.
- Fm
- Dark-adapted Maximal Fluorescent Yield reached with a saturating pulse of light.
- Fm’
- Light-adapted Maximal Fluorescent Yield reached with a saturating pulse of light.
- Ft
- Fluorescent Yield at a given time, generally just before a saturation pulse is applied to a sample.
- Fv
- Variable Fluorescence (Fm – Fo or Fm’-Fo’).
- Fv:m or Fv:Fm or dark-adapted yield
- Maximal Quantum Yield of a dark adapted sample and equals (Fm-Fo/Fm).
- qP (photochemical quenching)
- (Fm’ – Ft)/ (Fm’-Fo). Photochemical fluorescence quenching is indicative of the proportion of PAR absorbed by the ‘open’ reaction centers of PS II and hence used in photochemistry. This coefficient may vary between 0 and 1.
- Chronic Photoinhibition
- Photoinhibition is characterized by a type of non-photochemical quenching which recovers only slowly (if at all) in the dark.
- Dark Adaptation
- A brief (usually 30 minute) acclimation time in darkness. During this time, photochemical reactions stop, and all reaction centers ‘open’ to receive light energy when it becomes available.
- Dynamic Photoinhibition
- The same as NPQ: The quenching of fluorescence by dissipation of excess light energy as heat. Involves xanthophylls.
- Saturation
- Maximum photosynthetic rate or photosynthetic capacity.
- Thylakoid
- A lipid membrane within a chloroplast that contains photopigments comprising PSI and PSII.
- Yield (light adapted)
- Quantum Yield of photochemistry PS II, measured on light adapted samples. (Fm’-Ft)/Fm’ (or ΔF/Fm’).
- Yield (dark adapted)
- Quantum Yield of photochemistry in PS II, measured on dark adapted samples. (Fm – Fo/Fm).
- Zero Offset
- This number represents a background signal found within the instrument. Abbreviated as ‘Zoff’, it is automatically subtracted from Ft, and all consequently determined fluorescent values.
- qN (non-photochemical quenching)
- (Fm-Fm’)/(Fm-Fo) or, alternately, (Fm-Fm’)/Fm-Fo’). This coefficient may vary between 0 and 1. However, if qN exceeds 0.4 there is also significant quenching of Fo, and NPQ should be examined. Hence, qN = (Fm-Fm’)/ (Fm – Fo’). Note: This formula has also been used for qN (qN = 1 – (Fm’ – Fo’)/(Fm – Fo) = 1 – Fv’ : Fv), and provides values very close to that immediately above. Useful only when photosynthesis is activated, usually after 2 minutes of illumination.
- NPQ
- (Nonphotochemical quenching or Nonphotochemical exciton quenching – Kanazawa and Kramer, 2002). NPQ = (Fm-Fm’)/Fm’. NPQ can vary between 0 and infinity, but, for practical purposes, is unlikely to exceed a value of 10. The choice between NPQ and qN depends upon the application – with NPQ, that part of photochemical quenching is emphasized that reflects heat-dissipation of excitation energy in the antennae system. (Hence, NPQ is a convenient indicator of ‘excess light energy’ – Schreiber, 1997). NPQ is relatively insensitive to that part of non-photochemical quenching which is associated with qN values between 0 and 0.5. Nonphotochemical quenching of excitation energy, which protects higher plant photosynthetic machinery from photodamage, is triggered by acidification of the thylakoid lumen as a result of light- induced proton pumping, which also drives the synthesis of ATP. In essence, excess absorbed light energy is dissipated as heat within the light-harvesting complexes. NPQ
involves two processes activated by the acidification of the lumen, the interconversion of xanthophyll cycle carotenoids, and the protonation of residues on key LHC components. In absence of NPQ modulation, buildup of reduced electron carriers would block electron flow before the lumen could be significantly acidified. This over-reduction could result in the formation of a stable, doubly-reduced Qa species in PS II, allowing the formation of triplet chlorophyll species, which in turn can react with O2 to form singlet oxygen (1O2), an extremely toxic oxygen radical. - ETR (electron transport rate)
- Effective quantum yield (Fm’ – Ft)/Fm’ X PAR. Ralph, Gademann, Larkum and Kuhl (2002) believe Beer et al. (1998) underestimated absorption coefficients of corals (measured as 0.023 – 0.036, as compared to 0.86 for terrestrial green leaves). Hence, Ralph et al. recommend reporting ‘Relative ETR’, as determined by the above formula, until a widely accepted method of determining absorption coefficients is established.
- Xanthophylls
- Oxygenated carotenoid pigments produced by plants. Xanthophylls are anti-oxidants and may help detoxify oxygen radicals.
Some are also involved with energy dissipation which involves light mediated changes to their structures.
References
- Atkinson, M. and C. Bingman, 1999. The composition of several synthetic seawater mixes. Aquarium Frontiers Online. March, 7 pp.
- Atkinson, M.J., E. Kolter and P. Newton, 1994. Effects of water velocity on respiration, calcification and ammonium uptake of a Porites compressa community. Pac. Sci., 48(3):296-303.
- Atkinson, M.J., B. Carlson and G.L. Crow, 1995. Coral growth in high nutrient, low-pH seawater: a case study of corals cultured at the Waikiki Aquarium, Honolulu, Hawaii. Coral Reefs.
- Atkinson, M.J. and R.W. Bilger, 1992. Effects of water velocity on phosphate uptake in coral reef-flat communities. Limnol. Oceanogr., 37(2):273-279.
- Beer, S., M. Ilan, A. Eschel and I. Brickner, 1998. Use of pulse amplitude modulated (PAM) fluorometry for in situ measurements of photosynthesis in two Red Sea faviid corals. Marine Biology, 131: 607-612.
- Bongiorni, L., Shafir, S., Angel, D. and B. Rinkevich, 2003. Survival, growth and gonad development of two hermatypic corals subjected to in situ fish-farm nutrient enrichment. Marine Ecology Progress Series 253:137-144.
- Falkowski, P. and Z. Kolber, 1995. Variations in chlorophyll fluorescence yields in phytoplankton in the world’s oceans. Aust. J. Plant Physiol., 22: 341-355.
- Fitt, W. and M. Warner, 1995. Bleaching patterns of four species of Caribbean reef corals. Biological Bulletin (Woods Hole), 187, 298-307.
- Gorbunov, M., P. Falkowski and Z. Kolber, 2000. Measurement of photosynthetic parameters in benthic organisms in situ using a SCUBA-based fast repetition fluorometer. Limnol. Oceanogr., 45(1), 242-245.
- Gulko, D., 1998. Hawaiian Coral Reef Ecology. Mutual Publishing, Honolulu. 245 pp.
- Hall, D. and K. Rao, 1999. Photosynthesis. Cambridge University Press, Cambridge. 214 pp.
- Hoegh-Guldberg, O., 1999. Climate change, coral bleaching and the future of the world’s reefs. Mar. Freshwater Res., 50, 839-866.
- Jones, R., O. Hoegh-Guldberg, A. Larkum and U. Schreiber, 1998. Temperature-induced bleaching of corals begins with impairment of the CO2 fixation mechanism in zooxanthellae. Plant, Cell and Environment. 21, 1219-1230.
- Kanazawa, A. and D.M. Kramer, 2002. In vivo modulation of nonphotochemical quenching (NPQ) by regulation of the chloroplast ATP synthase. Proc. PNAS, 99(20): 12789-12794.
- Kirk, J.T.O., 2000. Light and Photosynthesis in Aquatic Ecosystems. Cambridge University Press, Cambridge. 509 pp.
- Kolbert, O. Prasil and P. Falkowski, 2000. Measurements of variable chlorophyll fluorescence using fast repetition rate techniques: Defining methodology and experimental protocols. Biochim. Biophys. Acta., 13 (67): 88-106.
- Ralph, P.J., R. Gademann, A.W.D. Larkum and M. Kuhl, 2002. Spatial heterogeneity in active chlorophyll fluorescence and PSII activity of coral tissues. Marine Biology, 141: 639-646.
- Schreiber, U., 1997. Chlorophyll Fluorescence and Photosynthetic Energy Conversion. Heinz Walz GmbH, Effleltrich. 73 pp.




0 Comments