Triton Lab ICP-OES Testing of a Certified Artificial Saltwater Standard
By Rich Ross and Dr. Chris Maupin
At MACNA 25 in Denver, the potential for the new ICP-OES aquarium water testing by Triton Lab in Germany made many saltwater hobbyists swoon. The possibility of getting a real good look at what is going on chemically with the water in our glass boxes for an affordable price is incredibly appealing, since traditional ICP-OES testing is expensive and time consuming. In this installment of Skeptical Reefkeeping, we explain how ICP-OES testing works, explore the challenges of testing saltwater with ICP-OES, develop an understanding of the methods used to address those challenges, and discuss the results from the testing of a certified artificial saltwater standard that was sent to Triton Lab.
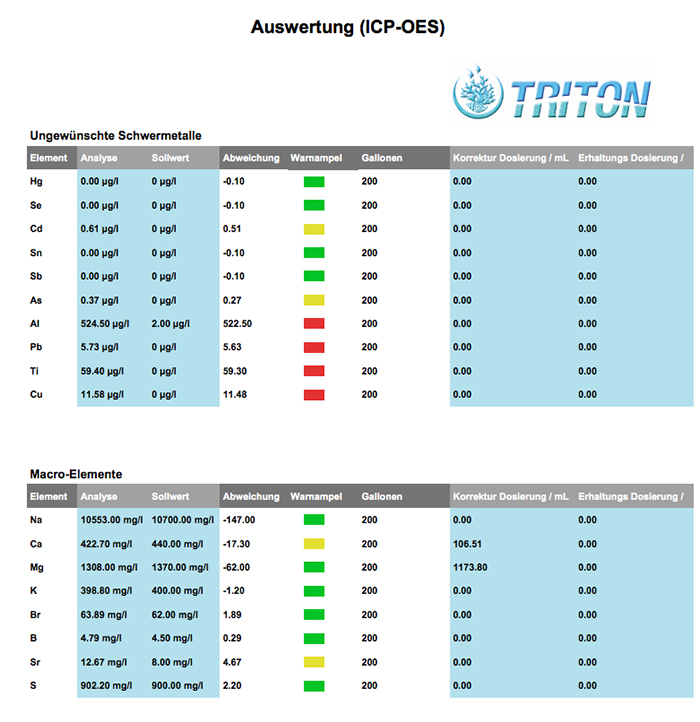
A partial look at some of the Triton Lab results taken from the available downloadable PDF. A discussion of the “Triton Method”, the “setpoints” used by Triton Lab, or what levels of which elements require action on the part of the aquarist is beyond the scope of this article.
A Brief Reminder to Set the Scene
Skepticism is a method, not a position. It can be defined as a method of intellectual caution and suspended judgment. As a Skeptical Reefkeeper, you decide what is best for you, your animals, and your wallet, based upon critical thinking, not just because you heard someone else say it. The goal of this series of articles is not to provide you with reef recipes or to tell you which ideas are flat out wrong or which products really do what they say they do or which claims or which expert to believe. The goal is to help you make those kinds of determinations for yourself, while developing your saltwater expertise in the face of sometimes overwhelming or conflicting advice.
Introduction: Why We Don’t all use ICP Testing Already
When reefkeepers daydream about water testing, they tend to imagine testing their aquarium water with a university or research institution’s complicated and expensive inductively coupled plasma (ICP) mass spectrometer or optical emission spectrometer, because, when properly calibrated and used, these instruments can produce incredibly accurate and precise results. However, a request to test tank water often is met with the response “Get that the hell out of my lab” from the lab technician, or by a seemingly outrageous price from a commercial lab.
Saltwater presents ICP testing with special problems. Most notably, it can be detrimental to the instrument’s components, and the saltwater itself can help produce unreliable results. Compensating for these issues makes the testing expensive and time consuming. Sometimes months are required to get results, and at considerable financial cost: sometimes hundreds if not thousands of dollars per analysis of each individual element. However, Triton Lab is now offering ICP OES testing of 321 elements in a saltwater sample for a reasonable cost and a fast turnaround2 – a service that no other ICP-OES lab seems willing to provide. In the Skeptical Reefkeeping series, we often quote They Might Be Giants – “Are you sure that that thing is true, or did someone just tell it to you”3, and in that spirit we looked at some of the Triton Lab claims and are sharing our results. It is important to note that that being skeptical is not the same as being cynical, and that nothing would please us more than an inexpensive, accurate and precise water test providing values for 32 elements in a single report at a relatively inexpensive cost; however, understanding traditional ICP-OES and the numbers from our experiment tell a more complicated story.
How ICP-OES Works
In chemistry class, many of us became familiar with a flame test, where you put some stuff in a Bunsen burner and watch what color the flame burns, indicating what elements are in the stuff you burned. Table salt, for example, turns the flame yellow/orange (fun fact – both authors suffer from poor color perception) due to the presence of sodium in the salt. This test is the beginning of many a mad scientist – Muhahaha! Colored flame! An ICP-OES, when distilled to its most bare fundamentals, is a sensitive, quantitative means of performing a flame test. Instead of a natural gas flame in a Bunsen burner, it uses an argon plasma torch to burn the ions and record the colors they produce, and how brightly, which tells us what elements in what amounts, are in the sample. Muhahaha plasma! The ICP in ICP-OES is the inductively coupled plasma, while the OES stands for optical emission spectrometer.

Looking inside the front end of an ICP-OES. Photo by Jennifer Flannery.
The Gory Details
Conventional ICP instruments use argon gas, swirling it in a helical fashion in concentric quartz tubes. A high power (typically approximately 1500 watt) radio frequency (RF) power generator is attached to a copper load coil that surrounds this quartz “torch.” This RF power causes an alternating current to oscillate within the load coil, producing RF magnetic fields within the inert argon gas. An initial spark capable of ionizing some of the argon is applied. Ionization means that free electrons are generated in the torch. These ions are then slingshot by the load coil fields, smashing and stripping other electrons and argon atoms (the “inductive coupling” part of ICP), creating and maintaining a plasma discharge. The process raises the temperature through collisions of argon atoms and ions and electrons. Typical analytical ICP temperatures are 7000K – 10,000K, and are appropriately a whitish blue color (fun fact – the surface of the sun is only approximately 5770K).
Atoms to be measured in a solution (seawater is considered a solution) are first nebulized into a fine mist using dry argon gas. These mist particles are then carried from the spray chamber into a ceramic injector that reaches into the plasma and strips away any remaining water molecules, leaving individual particles (this process is known as desolvation). These particles are injected into the hotter areas of plasma, where they become fully atomized. Subsequently, the electrons in the atoms undergo thermal excitation from ground state (the state of least possible energy) to the point where outer electrons are lost and the atoms become ionized. Remaining electrons return to the ground state from excitation and emit electromagnetic radiation in the form of photons of discrete wavelengths. Every electron returning to the ground state emits photons with characteristic wavelengths, with many being in the ultraviolet (UV) and visible spectra. This energy difference (between the exited and ground state) is related to the wavelength of photons emitted. Each element has a characteristic emission spectrum, which means that the photons are only emitted at certain wavelengths and these wavelengths can be used to identify the presence of an element and the intensity of the emission can be used to determine the concentration.
The photons are separated into distinct photon “beams” of different wavelengths. Once these wavelengths have been separated, the number of photons are then assessed and recorded for each wavelength via one of many potential collector types (photomultipliers, charge transfer devices, etc.). Calibrations, discussed later, relate these photon counts to actual analyte (what we’re measuring for) concentrations.
Five Challenges of Testing Saltwater with ICP-OES
1) Interference
One of the fundamental drawbacks from highly effective thermal excitation in a rich mix like saltwater is the likelihood of emission wavelengths from the different elements in the sample overlapping and causing interference in measurement. This interference can make the results of the testing for any particular element inaccurate. Ideally, many elements of interest are isolated from this brutal cocktail of dissolved solids by methods that are dependent on the element itself. The details of such techniques have formed the foundations of entire dissertations and scientific careers.
Since they are testing 32 elements at the same time, we are unsure how Triton Lab is dealing with this interference challenge. In online discussions and personal communication, we have been told that they are utilizing proprietary techniques. However, without knowing what techniques Triton Lab is using, we are unable to judge its effectiveness as compared to existing published methods and standards: no other lab has been able to affordably overcome this interference problem.
2) The Nebulizer
To nebulize and separate (desolvate) the atoms in the sample from the solvent (the solvent in this case is water), the solution must be pumped through a fine orifice, where it is turned into a cloud by the dry argon gas inside a spray chamber and carried into the ICP torch injector. The injector is typically a ceramic tube that passes the atoms into the plasma, where they are desolvated, ionized and excited. As a result of this function, the injector is prone to becoming clogged by solid build up of material in the sample. The more dissolved solids in the sample, the faster the build up. Since saltwater is chock full of dissolved solids, injecting saltwater often clogs the injector. In other words, the ICP gets salt creep, which plays havoc with the sensitivity of device, causing changes and drift in the analysis. Samples like saltwater with a high amount of dissolved solids require special techniques that take time and cost money to dampen solid build up in the torch injector. To make things more complicated, different nebulizer designs can be implemented to help reduce or slow this build up. The most prevalent design, the concentric nebulizer, however, is especially prone to this issue.

The nebulizer and spray chamber. Photo by Jennifer Flannery.
We have asked Triton Lab which nebulizer is being used and how the solid build up is being managed they have not answered this question directly – presumably for proprietary reasons – and we are therefore unable to comment on what influence, if any, it may have on the Triton Lab results.
3) LOD – Limit of Detection
The limit of detection (LOD) is expressed as the concentration derived from the smallest quantity of an analyte that can be detected with a reasonable certainty for a given analytical procedure. A common misconception of the LOD is that it defines the smallest concentration that can be measured. However this is not the case, it defines the smallest concentration at which we can decide if the element is present or not. In other words, being able to distinguish a signal from the background noise.
To determine LOD a “blank” solution is used. For aquarium saltwater, an ideal blank would be an otherwise complete seawater matrix with the element of interest (e.g., iron), completely absent. Several samples of the blank solution are analyzed. The mean and standard deviation of the blank background noise is recorded at the wavelength of the element of interest. The smallest measure of the element of interest that can be detected with reasonable certainty, is three times the standard deviation of the blank background noise
This “smallest measure” is converted to concentration via the Sensitivity S, which is defined as the change in concentration divided into the change in counts per second of light striking the instrument detectors (slope of the calibration curve). Since the concentration in the blank is zero, the concentration of the element is three times the standard deviation of the blank times the slope of the calibration curve. This concentration is reported as LOD.
Triton Lab reports LOD as a range. When asked about this in online discussions and personal communication, we were told that this is because of difference in calibration and that different calibrations will result in different LOD’s. The concern we have about this is that the aquarist doesn’t know which calibration is being used at the time at which their test was done. If the test result falls between the LOD range given, it may be below the actual LOD which means we cannot determine if the element is present or not. See below for a discussion of concerns regarding how elements below Triton Lab LOD are expressed.
4) Calibration
Calibration is how the counts of light per second are translated to concentration values of the element of interest. At some level, there are nonlinearities in calibrations, despite extremely high correlation coefficients in calibration curves reported by Triton. Calibration curves should be generated using solution concentrations to bracket expected values of the element of interest as closely as possible. The ideal way to do this would be 4 calibrations for each element to be tested. The first calibration should be the “matrix blank”, the matrix being the solution you are testing (in this case seawater), which would have everything that would normally be in the matrix except the element for which you are calibrating. The second, third and fourth calibrations should be the matrix (again seawater in this case) with the element of interest at low, medium, and high concentrations that bracket the expected concentration value of the solution to be analyzed. This allows nonlinearities that arise from matrix and overlap issues to be accounted for properly and will ultimately lead to more accurate results. Calibrating for all the elements in seawater at the same time usually leads to incomplete accounting for potential matrix interference (see Interference above).
According to “TRITON ICP – accuracies and limits of detection V 1.1”4 – “We carry out a full calibration on our ICP-OES every day before testing and after every 30-40 tests.” For the small amount of money being charged for testing it seems improbable that each element is being calibrated individually. Since we don’t know the methods of calibration Triton Lab is using, as we are told they are proprietary, we don’t know that Triton Lab calibration appropriately accounts for the drift and interference present in all ICP instruments.
5) Alpha and Beta Error
During actual testing, if enough photons reach the detector to surpass the LOD, there is a 1.0% chance of this detection being a false positive. This is referred to as the alpha error (see Figure 1). However, because the distribution of the light counts measured in the blank and the element tested overlap at the lower end of the LOD distribution there is statistically a 50% chance for false negative. This is referred to as the beta error (See Figure 1). In true analytical chemistry, the LOD is typically referred to only in cases where a binary, qualitative answer is required: Is the element of interest present, or is it not? If yes, then you can be confident (99% confident) that it is not a false positive. However, if the element appears to be absent, keep in mind that your odds of it being a false negative are the same as a coin toss! There is a 50/50 chance that the element is still present, and you cannot know by the very definition of the LOD!
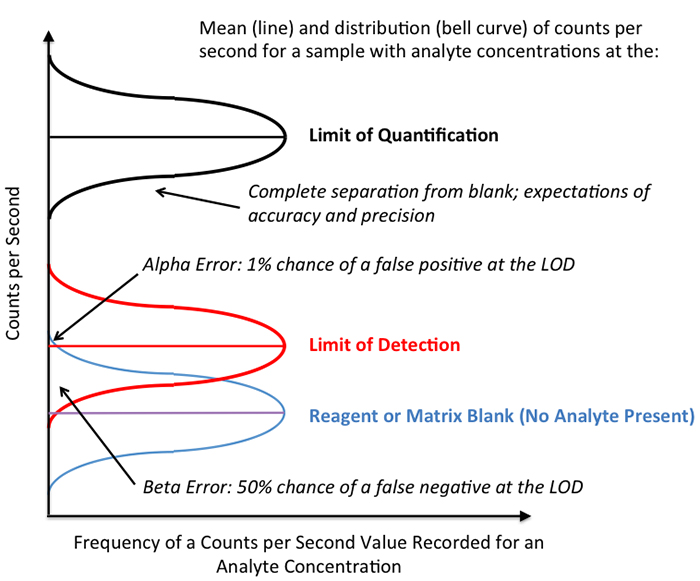
Figure 1. Schematic representation of an reagent or matrix blank, a sample with an analyte concentration at the LOD (as defined in the text as 3 times the standard deviation of the distribution of noise in the blank), and at the LOQ.
To actually quantify photon counts into meaningful concentration values, relating them to how much of the element is present in the solution, there must be enough of the element present to reach or surpass the limit of quantitation, or LOQ. The LOQ is the minimum amount of the element that must be present to be quantified within specified accuracy and precision limits. Often, the LOQ requires anywhere from 2 to 10 times more of the element to be present than the LOD value, but it is dependent on each matrix, element, and OES instrument, and it must be critically assessed by a skilled analyst.
So why does this create a noteworthy issue with respect to ICP-OES and seawater? To achieve measurable concentrations, many elements require preconcentration, or artificially increasing their concentrations in solution sometimes by an order of magnitude or greater. Many preconcentration and coprecipitation (the removal of a particular element from the matrix for independent quantification) techniques have been developed in earnest for specific elements, and viably remedy this issue, or work around it. Ultimately, however, many trace elements simply require more sensitive or specific techniques, which are beyond the scope of our discussion here, to accurately measure the concentration of certain elements in seawater.

Venn Diagram of Precision and Accuracy. To have both, you want the mittens on the kittens. Photo by Google Images.
From online discussions and Triton Lab documentation, it appears that Triton Lab does not preconcentrate or coprecipitate samples to be tested, and that results for all elements are determined at the same time by a single processing of the sample. It is also unclear how LOQ is being determined or if the alpha error is being considered. It is important to note that because of beta error, when Triton Lab reports a value of 0.00 for a particular element, depending on the photon counts at the wavelength of interest actually being registered by the detector, there may be a 50/50 chance that that element is actually present.
Testing the Test
Triton describes their services by saying “The TRITON water analysis is the first and most accurate trace analytic laboratory service for marine hobbyist and professional aquarists,”5 The Triton US Facebook page elaborates by saying “Triton is a highly accurate, professional product with a sophisticated scientific pedigree.”6 However, the issues we have outlined above make it difficult to judge and assess the validity of these claims as Triton Lab methodology seems to be different from any other methodology being used today. We don’t know what proprietary techniques are being used or how well they work, we don’t know what kind of nebulizer is being used or how it influences results, there are LOD and Alpha error as well as calibration concerns. In the absence of the knowledge to make informed assessment of the ability of the Triton ICP-OES to deliver as claimed, the only other alternative left to us is to run some blind tests through Triton Lab using known standards and methods, and to compare the results against the known standards.

Page one of certificate of analysis of the saltwater standard sent to Triton Lab. We recommend that Triton conform to international standards by assessing the mass of samples, eliminating density, and therefore weight/volume concentration units of the sample as a cause for concern in the accuracy of Triton’s results.
An artificial seawater standard was obtained from High Purity Standards Certified Reference Material – Seawater (CRM-SW)7 and sent to Triton Lab for testing. This standard is prepared volumetrically and gravimetrically, and is intended and designed to be used to compare analytical values generated by testing devices like ICP-OES. The amounts of the different elements in the standard are accurate, precise and certified, so running the standard through the Triton Lab process allows us to assess the accuracy and precision of the Triton Lab testing.

Page two of certificate of analysis of the saltwater standard sent to Triton Lab.
It is important to note that the certified artificial seawater standard has been acidified such that it contains a final composition of 2% HNO3 (nitric acid) by weight. This acidification is standard operating procedure for preparation and storage of natural waters and standards, as it is considered a requirement to prevent loss of analyte from solution (in this case, the 32 elements to be tested) primarily through adsorption to solution container walls. The 2% nitric acid in the certified artificial seawater standard has no meaningful impact on ICP-OES test results (see Figure 2).
The standard was separated into three identical samples, and each submitted to Triton Lab as separate samples to be tested individually. We did not inform Triton Lab about the samples beforehand, which ensured that the samples of the standard would not get any special treatment or be treated differently from the other eight samples of aquarium water that were sent at the same time.
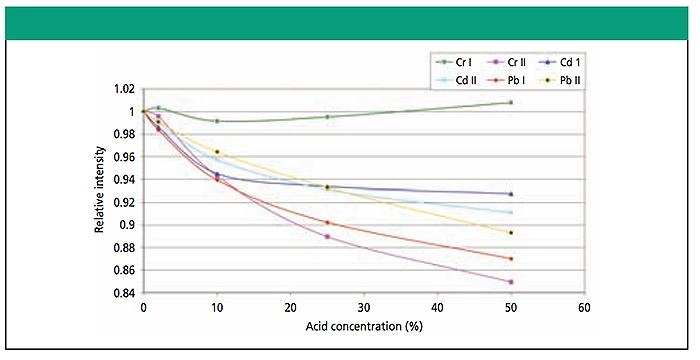
Figure 2. Effect of nitric acid concentration. The addition of 2% nitric acid has negligible effects in ICP-OES testing, as shown from this figure from “Matrix Effects in ICP-AES Analysis” (Sivakumar, et al)12. Also see “ICP-OES Determination of Select Metals in Surface Water – a Metrological Study” (Chochorek et al)13 for further discussion of acidification of samples.
In personal communication with Triton Lab and Triton US, we have been informed that Triton Lab does not acidify their standards used for calibration because they claim “samples not sent in seawater will not test accurately”, nor are samples to be tested acidified prior to shipping (fun fact – Like many things in this hobby that have cautionary Safety Data Sheets like salt mix, calcium hydroxide, vinegar, test kit reagents, salt water, etc, samples acidified in this manner do not constitute “hazardous” hazardous materials); since Triton Lab seems to be the only lab testing in this manner, we cannot comment on this non-acidification because we are not aware of any method that forgoes acidification while still claiming to maintain the integrity of analyte concentrations in solution.
Analysis
For this discussion, we need to differentiate between precision and accuracy8. Accuracy is how close the result is to the actual value of the sample, and precision is how close multiple tests are to each other. Usually, when testing aquarium water, accuracy is almost always an unknown, and you are relying on the test itself to give you a value. In this case, with the CRM-SW sea water standard, we know the actual values of the each element in the standard, so we can determine the accuracy of the test. Since we had three samples of the standard tested, we can also estimate how precise the testing method is.
Accuracy
As an example, the iron value (see Table 1) in the CRM-SW standard is 21 µg/l, and the average of the three measurements performed by Triton Lab of this reference standard is 41.23 µg/l. Assuming a Normal Distribution of data, the two-standard deviation distribution of the three analyses was ±1.6 µg/l. In other words, if we were to measure our reference material a fourth (or fifth, sixth, etc.) time with the Triton testing, there is a 95% chance that the value will fall between 39.63 and 42.83 µg/l. Note that the standard value of 21 µg/l is outside this range. This also means there is a greater than 95% chance that any measurement will not overlap with the known value of the standard. Thus, the reported results will not be accurate by the definition of accuracy: that the measured value and the known value are indistinguishable within the limits of uncertainty of the method. In our case, this uncertainty is estimated by two times the calculated standard deviation of the three replicates of the instrument.
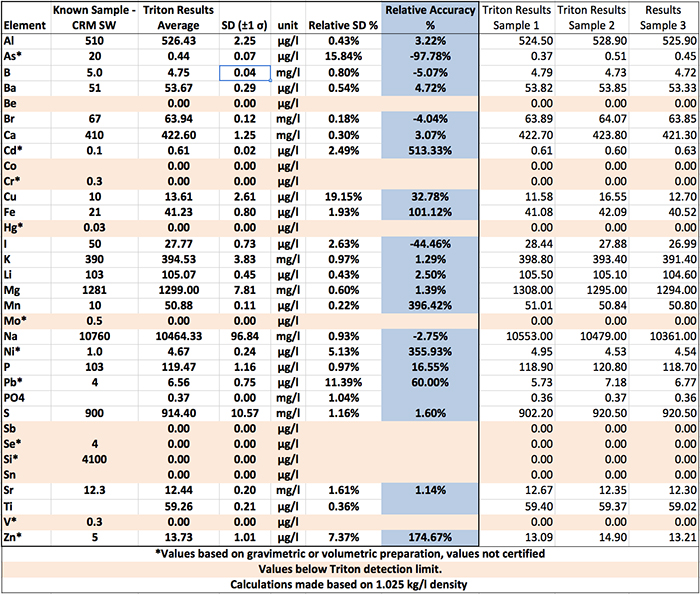
Table 1. Triton Lab test results and analysis. The CRM-SW reference values are reported by certification in units of weight of element over weight of solute (seawater in this case), for example, the mg of Sr in a kg of seawater. CRM-SW reference values require a conversion using an assumed density of 1.025 kg per liter to make the CRM-SW contents directly comparable to the Triton results. We report the CRM-SW concentrations with the appropriate number of significant figures.
Strontium is a good example of an element that produced one of the most accurate results in our experiment (1.14% relative accuracy), where the reference material value falls within the distribution of the Triton data, but we should take a moment and look at one of the most inaccurate results. The manganese value (see Table 1) in the standard is 10.3 µg/l, and the average of the three measurements performed by Triton Lab of this reference standard is 50.88 µg/l, with a 95% chance that any measurement of manganese will fall between 50.66 and 51.10 µg/l, again based on the distribution of the three replicates. In relative terms, the Triton test returned a result for manganese inaccurate by (difference between known and measured values, divided by the known value) 396.42%.
Precision
The precision in this context is the quantification of the distribution of values upon repeated analyses of the same sample, and is independent of accuracy. In regards to our experiment with the saltwater standard, the standard deviation divided by the mean gives us a distribution of the measured values relative to the mean amount of each element reported by the instrument (also called relative standard distribution). In the case of this Triton Lab result for strontium this relative standard deviation is 1.61%. This is a fairly common ballpark specification for precision (1%), so here the instrument performs, as it would be expected to perform. The other elements tested by Triton Lab fall mostly within the same precision, so the relative precision of Triton Lab testing for strontium is good.
The detailed results for the different elements are shown in Table 1. As seen from the results, the relative accuracy and relative standard deviation range vary considerably for different elements. For instance, consider copper in contrast to the above example of strontium, where both accuracy and precision are excellent. Copper tends to be a concern for marine aquarists in general, so it would be a boon to have both accuracy and precision for this element. The relative standard deviation for copper in the three replicate measurements is 19%. The exact same solution gives measured values with a range of 5 µg/l. The average of the three measured values is 13.6 µg/l, whereas the certified concentration of the standard is 10 µg/l. This is an example of an element that actually is accurate within the precision of the Triton Lab instrument – but only because the precision of the instrument is large enough that a Normal Distribution of measured values for copper encompasses the true, certified value, based on our 3 measured replicates. If we only had a single measurement instead of three, we couldn’t be sure how large the distribution was, nor would we be able to comment on the precision of the instrument.
“Fitness for Purpose” – A Hobby Gray Area
In online conversations of using Triton testing ICP-OES for reef aquariums, the idea of “fitness for purpose” was expressed as selling point. The issues outlined above regarding using ICP-OES for testing many elements in a single, non-concentrated test and limits based on LOD were discussed. After discussions of proprietary methods for dealing with those problems (which we problematically don’t know about) were tabled, the idea that even if Triton Lab testing wasn’t going to be as accurate and precise as the normal ICP testing, it was still good enough for reefkeeping, and that the Triton Lab testing was “fit for our purpose”. The reality, as it so often is, is more complicated.
According to the Triton US Facebook page, “Triton is a highly accurate, professional product with a sophisticated scientific pedigree. It’s not part of a grandiose thesis-level research product. Although it utilizes some of the most sophisticated technology available, it’s not intended to be utilized in academic research. Rather, it’s designed for professionals and hobbyists who would like a higher level of understanding about the composition of their reef aquarium’s water, with very sensitive limits of detection.”9 This kind of language makes our skeptical alarm bells start to ring. If Triton is highly accurate and professional with a sophisticated scientific pedigree, we have a hard time understanding why its results are only fit for purpose for reefkeeping – especially if those reefkeeping purposes are described as needing very sensitive limits of detection. Later in the same piece, Triton US says that “Triton’s testing has been utilized in practice at public and private aquariums all over the world for the past 7 years- a testimony to the accuracy and usefulness of this product” which also makes our skeptical alarm bells ring. Not only is the research done at public aquariums often academic research published in peer reviewed journals, but the last part of the statement is fallacious10 as the use of a product does not testify to its accuracy. People using a product simply means that people are using it, not that it actually does anything; lots of people may use ginger or bananas to cure ich, but that doesn’t mean that ginger or bananas actually cure ich11 (fun fact – ginger and bananas do not cure ich).
Marketing points like the above aside, are the results of the Triton test fit for our purposes? The answer to that seems to be “maybe” and “it depends.”
The strontium value in the SRM-SW standard is 12.3 mg/l, and the average of the three measurements performed by Triton Lab of this reference standard is 12.44 mg/l, and your initial thought could, and should, be “that’s really pretty close!” Whoo hoo, right? Hold on a sec. The manganese value in the standard is 10 µg/l, and the average of the three measurements performed by Triton Lab of this reference standard is 50.88 µg/l. Your initial thought regarding Manganese could, and should be, “That’s not close at all!” Boo hoo, right? Whoo hoo or boo hoo seems to be element specific in regards to the Triton Lab results we received.
So Where Does that Leave Us?
It is our opinion that some element results of the Triton Lab ICP-OES testing may be fit for our purposes, or may be close enough as to be considered fit for our purposes, while others may not. We suggest that readers go through Table 1 and take a look at the relative accuracy of each tested element and make their own decisions. The conclusion of the thought experiment on the strontium data is that the Triton instrument is pretty good at capturing the same elemental values on a sample measured multiple times. This is a good thing. However, the accuracy of the elemental measurements is lacking. We think that Triton testing might be useful for looking for trends or for pointing out large excesses or lack of a particular element known to be problematic, but that using the results to fine tune trace element levels may not be feasible.
It should also be noted that our three tests were done at the same time, which means we really only know the precision and accuracy for that one day, and that on other days when the specifics of the ICP-OES instrument are different, the relative precision and accuracy may be different. More replicates over time would build a clearer picture of the distribution of Triton Lab results.
It is also important to point out that Triton seems to list results that are below the instrument’s LOD as 0.00, which may be misleading. There are levels between 0.00 and Triton’s lower limit of detection (these vary depending on the elements), but the instrument cannot read those low levels. Additionally, we worry that listing results below the LOD as 0.00 might mislead aquarists to think their levels are actually 0.00 when in reality it is unknown what the levels are. Changing 0.00 to something like ND (not detected) on the results might help avoid this confusion. There are also alpha and beta error issues to consider.
Standard deviation calculations can be easily performed in spreadsheet software, and you can perform these exercises for any of the reported CRM-SW results presented here.
Fun fact – both authors would like nothing more than Triton Lab proprietary ICP-OES methods to be published, repeated, and accepted because such methods, once verified, could turn what we currently know about ICP-OES testing on its head. Inexpensive, easy, accurate and precise testing would be a huge benefit to the hobby and the scientific community at large.
We encourage you all to think critically and skeptically regarding Triton Lab testing results and marketing claims, and this article.
Thanks to Craig Bingman, Sanjay Joshi, Jennifer Flannery, all the participants in online discussions, Triton Lab and Triton US for their help in preparing this article, and a Special thanks to Jim Welsh for his amazing and incredibly helpful proofreading abilities.
Notes
- Phosphate is a calculated result, not a tested for result, so only 32 elements are tested for by Triton Lab
- http://www.triton-lab.de/index.php?id=25&L=1 $49.00 – http://triton-us.com/
- “Put it to the test” from the album “They Might be Giants – Here comes Science”
- TRITON ICP – accuracies and limits of detection V 1.1
- http://www.triton-lab.de/en/triton-lab/
- http://www.reef2reef.com/forums/triton-us/175064 and https://www.facebook.com/tritonLA/photos/
- http://www.highpuritystandards.com/
- See Skeptical Reefkeeping 9 for a longer discussion of precision and accuracy
- http://www.reef2reef.com/forums/triton-us/175064 and https://www.facebook.com/tritonLA/photos/
- See Skeptical Reefkeeping 1 for a discussion of fallicies
- See Skeptical Reefkeeping 1 for a discussion of ich treatments
- http://images.alfresco.advanstar.com/alfresco_images/pharma/
- http://www.pjoes.com/pdf/19.1/59-64.pdf



0 Comments