
Coral identification is a challenge for every aquarist, and few groups pose as many difficulties as the “closed brain corals”. Well known aquarium references (Sprung 1999, Borneman 2001) are rife with erroneous generalizations and outdated taxonomy. Retailers and wholesalers are often unreliable, and it’s generally best to ignore any of their identifications entirely. Quality field guides for the Indo-Pacific are almost nonexistent. Even the familiar scientific literature (Veron, 2000) can’t be relied on, as molecular studies have thrown these old, morphology-based classifications out the window. In sum, this is a confusing group with few reliable resources.
The term “closed brain coral” is used by aquarists with little thought as to what it precisely refers to. As commonly interpreted, the name signifies a couple things: 1) the coral in question is typically encrusting or massive 2) the coral in question has relatively large polyps, which may or may not chain together into meanders that resemble the folds of a brain. For decades, taxonomists agreed with this scheme and placed these species together in the family Faviidae. And so “closed brain” became a convenient term which obviated the need for aquarists to learn how to identify the bewildering diversity of morphologically similar corals that occupied the family.
But in the last decade a series of molecular studies have consistently shown that our traditional classification for these corals was enormously incorrect. Familiar groups like Favia and Montastraea are now known to form separate lineages split by ocean basin, and these two groups are so fundamentally different from each other that they necessitated placement into separate families. Montastraea was shown to be a particularly hot mess, with no fewer than six distinct genetic clades from four distinct families lumped under the taxon! Oulastrea, a genus most aquarists would confuse with Favia, turned out to be so distantly related to it that their evolutionary divergence likely predates the first appearance of the dinosaurs. So clearly, “closed brain coral” is a term that limits our ability to understand the fascinating evolutionary story these corals have to tell.
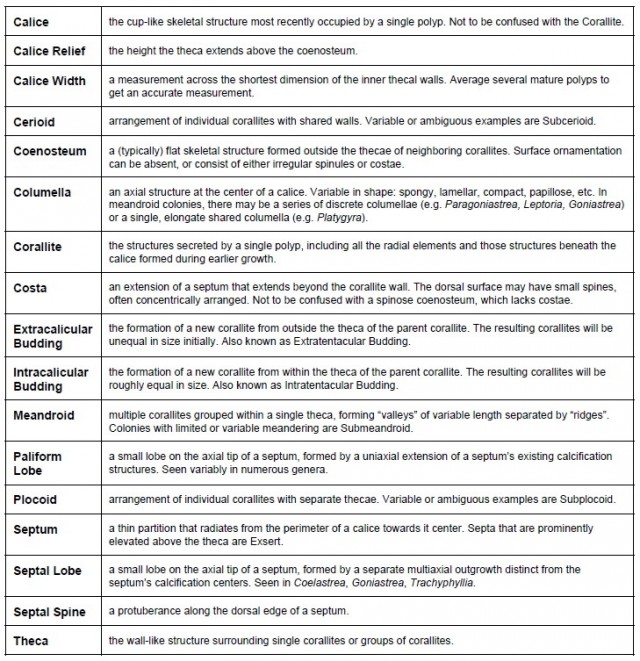
Now that a thorough taxonomic revision has appeared (Huang et al 2014), it’s time to review the current standing of these corals, and, most importantly, how we as aquarists can identify them. The latest taxonomic shuffling has added clarity to our understanding of how these species are related, but it has also added a slew of new scientific names. Some well-known genera, familiar to aquarists, divers and scientists alike, have had to receive new names in order to fit the molecular findings within the rigid system that governs zoological nomenclature.
The challenging situation that today’s researchers find themselves in is that most coral genera were first described in the nineteenth century, an era when scientists often had a poor understanding of the important features necessary for delimiting a species or genus. This resulted in many inadequate species descriptions, which is exacerbated by the fact that several of the original holotype specimens (which ultimately define what that species is) are now lost. Even those holotypes that remain can be ambiguous in their identity given the morphological plasticity present in corals, and none of these skeletons have any soft tissues with which to sequence DNA.
Much work yet remains before we fully comprehend the extent and limits of coral diversity and their byzantine evolutionary interrelationships. Particularly at the species level, our ability to delimit and diagnose one group from another is often based on an imperfect and tenuous understanding, which leads to “species” of questionable biological significance. Are Astrea rotulosa and A. devantieri actually two separate species, or is one just an artifact of taxonomic confusion? And what about Dipsastraea amicorum and D. mirabilis? How can we possibly know whether any of these “species” might be in danger of extinction if we can’t even agree on what they are?
Aquarists are generally quite far removed from the minutia and nuance of zoological classification and often fail to appreciate its ever-changing nature. The species to be discussed in this article exist as we know them today, but with more molecular study, many of these names will be combined or split into new species concepts. This article is an attempt to bring us as aquarists up to date with the latest scientific findings, but take it all with a grain of salt, as we still have much to learn.

Favia fragum & gravida. Credit: Florida Fish & Wildlife Conservation Commission & Pedro Milet Meirelles
Mussidae: Favia
The name Favia has traditionally been applied to a large, circumtropical group of corals that is commonly exported for the aquarium trade. This group was originally described (somewhat inaccurately) as possessing species with plocoid polyps and intracalicular budding. Molecular study has shown this to be erroneous, as two distantly related groups had been combined under this name: Atlantic “Favia” and Pacific “Favia”. This of course raises the question of which group is the true Favia, nomenclaturally speaking.
The answer isn’t necessarily straightforward, and there is disagreement amongst some of the taxonomic authorities on this matter. As it currently stands, the Atlantic species are now the only true Favia. It seems unlikely that aquarists will suddenly abandon their usage of this name en masse, especially since CITES permits will still require the old name Favia to be used for now. What we’ll likely see is something akin to how the name “tetra” is used amongst freshwater aquarists—a common name originating from long abandoned taxonomy.
These two groups, Atlantic Favia and Pacific ex-Favia, are easily separated by the shape of the spines on their septa. Favia, like the rest of the Atlantic brain coral family Mussidae, have perpendicularly arranged paddle-shaped teeth, while the Pacific Merulinidae have irregular, multiaxial spines. This might be difficult to visualize, so try to picture a septal spine shaped like an open hand versus one shaped like a wobbly cactus. These subtle differences were easy for generations of coral taxonomists to overlook, but molecular data has finally shed light on the extensive convergent evolution seen in the stony corals.
There is much debate concerning how many species are present in the Atlantic. Authorities like Veron treated fragum (=ananas) and gravida under one name, but recent molecular study indicates there are at least two distinct species. F. fragum and gravida are further distinguishable by the greater number of septa (with higher teeth) and greater calice relief in fragum. A third cryptic species, conferta, has also been suggested for submeandroid specimens, but its status as a valid species is contentious.
In their Atlantic range, there is little that would be confused with Favia. The overall shape is a small, hemispherical or encrusting colony with elongate or submeandroid polyps, which either lay flat or protrude strongly. Coloration is brown.
Merulinidae: Dipsastraea
The former Indo-Pacific “Favia” have now found a new home within the large family Merulinidae as Dipsastraea. With 22 extant species, this is the largest genus of merulinid coral, and it is hugely important in the construction of coral reefs. Unsurprisingly, it is also one of the most commonly exported corals for aquarists—trade names include: closed brain coral, pineapple coral, moon coral, etc. Identifying Dipsastraea is challenging, as the genus can vary widely in morphology between and within species.
Polyps are mostly medium to large in size and are highly variable in calice relief. Budding is primarily intracalicular, though at least some species (e.g. matthai, laxa, helianthoides) can form extracalicular buds as well. Polyps vary from highly plocoid (with large amounts of coenosteum separating the polyps) to subplocoid. The differences in polyp shape are heavily influenced by environmental conditions, with colonies originating from deeper, mesophotic reefs or calm lagoons showing a tendency towards decreased calcification, more widely spaced polyps, and a correspondingly more-plocoid appearance. Conversely, colonies experiencing heavy water flow become more heavily calcified, often with fusion of the thecal walls, and can thus strongly resemble the cerioid skeleton of Favites. This environmental plasticity in corallite arrangement is a trend seen throughout the groups to be discussed, and adds a frustrating layer of ambiguity that must be dealt with when making identifications.
There is still uncertainty regarding the precise limits of Dipsastraea. The molecular work of Huang et al 2014 indicates that both Coelastrea and Trachyphyllia may belong in this group, but the authors go on to mention that there are notable morphological differences, and that the genetic relatedness may be an artifact of the limited number of genes studied to date. Furthermore, the relationships amongst the currently recognized Dipsastraea species are poorly understood, and it is quite possible that the group could be further split upon future study.
Barabattoia is now regarded as a synonym by Huang et al 2014, who recognizes three species. These differ from other Dipsatraea by possessing particularly tall, plocoid polyps, with some specimens forming unusual subdendroid branches. These corals do occasionally turn up in aquarium stores, generally going unnoticed, unidentified and unappreciated due to their rather drab coloration. Wijsmans-Best reports this group to be found exclusively in sheltered habitats, with the tall calices presumably an adaptation to avoid sedimentation.
Distinguishing Dipsastraea from Favites is complicated by the general overlap of morphological characters amongst these two superficially similar genera. Remember that phylogenetic trees produced from molecular study show these two groups to be rather distantly related, so their similarities are the result of convergent evolution. Those specimens which are ambiguously plocoid/cerioid will be nearly impossible for aquarists to identify, especially if only a single living colony is available for study. Comparison of a series of specimens collected from varying habitats within the same region is generally required to conclusively reach an accurate identification. This is likely a frustrating revelation to many aquarists, as those in the hobby often seem to have an insatiable desire to attach a binomial to their purchases.
Fortunately, a large portion of specimens seen for sale are more clearly representative of their genus. Those specimens with clearly separated thecal walls and intracalicularly budded polyps are more likely than not Dipsastraea, and those specimens showing shared thecal walls are presumably Favites (or one of the genera resembling it).
One of the great challenges of identifying Dipsastraea (and most merulinids), is that environmental variations in water flow, light and sedimentation can cause fundamental changes to the skeletogenesis of the colony. A colony of D. favus from the turbulent waters of an upper reef slope can have tightly-appressed, short polyps with numerous septa and thick walls, whereas specimens from a silty bay or a deep reef can show tall, widely-separated polyps with fewer septa and thinner walls. These two forms can look like completely different species, and it’s only when studying a series of specimens spanning the entire range of ecological morphotypes that any sense can be made. There is still much disagreement about the limits of many of these species, with little genetic study to back up the current species concepts. For instance, a study of Favites (=Montastraea) valenciennesi indicated the presence of a second cryptic species that likely belongs in Dipsastraea, and it’s quite likely that some species currently recognized may turn out to be mere variants of another more heteromorphic species. Another molecular study (Arrigoni et al 2012) has shown that Acanthastrea faviaformis belongs within Dipsatraea, and the upcoming revision of the Lobophylliidae will presumably make the formal taxonomic move for this misplaced species.
Most Dipsastraea have relatively uninspired coloration, but a couple of exceptions have acquired their own trade names. Specimens with a tricolored appearance of a bright, marbled oral disk, dark walls and a bright thecal apex have been dubbed the “prism favia” or “dragon soul favia”. While many retailers have claimed this to be “Goniastrea palauensis”, the correct identification appears to be Dipsastrea rosaria, which Veron mentions as being distinctively colored in the species description. Also note that a similarly patterned coral, the “reverse prism favia” is likely a different species, possibly Favites flexuosa. This coral has a consistent color pattern and lacks the marbled oral disks of rosaria.
The second commonly encountered trade name is “Christmas Favia” or “X-mas Favia”, which is applied to any “closed brain” with red polyps on a green coenosarc. As we’ll see elsewhere in this article, there are at least three genera (Dipsatsraea, Favites, Astrea) and multiple species with this color pattern. Check the polyp shape and budding when identifying these corals.

The brief accounts in the above table will provide some assistance towards making determinations of species that might appear in aquarium stores. A few Indian Ocean endemics are excluded. Consult with the written descriptions and photographs in Veron 2000, Veron et al 1977, and Wijsman-Best 1972 & 1976.














Hi,
You can get 20 -50 Frеe LIKЕS
Visit —> { SOCLIKES COM / Tryit}