The food chain of a marine ecosystem can be better understood with the use of stable isotope analysis. An isotope is an atom with an abnormal number of neutrons. Isotopes exist in two forms: stable and radioactive. Radioactive isotopes decay over time, while stable isotopes do not, hence their name. Now that we are somewhat familiar with stable isotopes, I will attempt to explain how they may be analyzed to help us understand the food chain of any given marine ecosystem.
All plants and animals contain stable isotopes. These isotopes bioaccumulate in individuals as they consume prey items over time. Knowing that primary producers such as algae have very low concentrations of stable isotopes, an algivore will accumulate stable isotopes at a relatively low rate. The predator of this algivore, however, will accumulate stable isotopes at a slightly higher rate due to the more significant presence of stable isotopes within its algivorous prey. This trend applies all of the way up to apex predators such as toothed whales, polar bears, and sharks, which consequently exhibit the highest levels of stable isotopes.
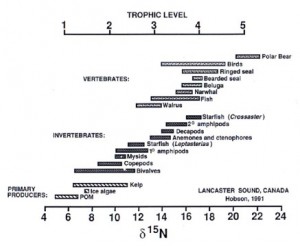
Hobson, 1991
Using a mass spectrometer, one may determine the stable isotope concentration of a tissue sample derived from a marine organism. The results are likely to provide a general understanding of the organism’s position in an ecosystem’s food chain.
Though in its relative infancy, stable isotope analysis may help us better understand coral reefs and the organismal interactions that drive their existence.










0 Comments