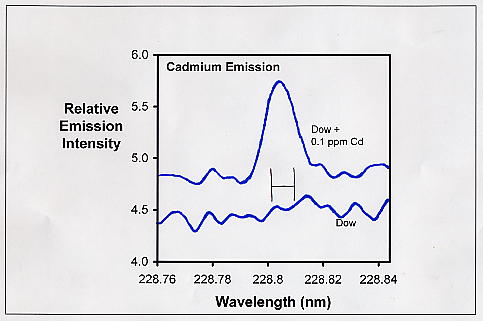
Phosphate is an ion of great concern to reef aquarists. In fact, aside from calcium and alkalinity, it is probably the chemistry topic on which reef aquarists focus the most. Much of this concern is warranted, with phosphate potentially contributing to algae problems, poor coloration of corals and other invertebrates, and growth of most photosynthetic organisms.…