
When you visit a local fish store to run some errands, you may stumble upon corals labelled as “curled, spiral or pigtail gorgonians”. In most cases, however, these specimens turn out to be antipatharians, also known as black corals. As these corals often have red, orange or yellow tissues, with no sign of black anywhere, one can understand the confusion. To the casual observer, black corals and gorgonians do look alike, and mixing them up is a common mistake in the aquarium trade. The confusion lies in the fact that the name “black coral” stems from the shade of the coral’s skeleton, not its tissue. In fact, most black corals do not appear black on the outside, although species exist that do. What gives black corals away quickly though is their polyps; these always bear six tentacles, in contrast to those of gorgonians, which always have eight. In addition, curled gorgonians are rare (an example is the genus Viminella), so most spiral-shaped corals in the aquarium trade are in fact antipatharians. This distinction between black and gorgonian corals is important, because they are not closely related and have a completely different anatomy.

Left: a yellow Cirrhipathes sp. growing in an aquarium. Right: the same specimen with its tissue removed, showing the typical black, spiny skeleton (right image composed of six stacked photographs). Photo by Marzia Bo and Tim Wijgerde.
History and Systematics
At present, black corals are placed in the order Antipatharia (phylum Cnidaria, class Anthozoa, subclass Hexacorallia), which consists of approximately 7 families, 42 genera and 230 species. Despite the relatively small number of species, black corals are spread throughout all the oceans of the world. They are found especially in tropical and subtropical areas, where they can reach very shallow waters, and in temperate and polar regions, but here never shallower than 167 feet (50 m) in depth. Often dwelling in deep waters, antipatharians remain poorly understood, both from a taxonomic and ecological point of view. This is also true for many other deep coral fauna, such as members of the Stylasteridae (Hydrozoa class), and the genus Gerardia.
Black corals are relatives of stony corals (order Scleractinia), amongst others, which also have polyps with six tentacles (or a multiple thereof). It took quite some time before biologists arrived at the current classification-the identification and grouping of living organisms-for black corals. The taxonomic origin of black corals dates back to the 18th century, when the German scientist Peter Simon Pallas separated antipatharians from gorgonians, as proposed by Linnaeus in 1758. He based this decision on their spiny skeleton and their coenenchyme (common tissue between polyps), lacking sclerites, and called them Antipathes. In Latin, the term Antipathes refers to a stone which was once thought to act as a charm against witchcraft. In fact, black corals were collected in ancient times for their magical and curative properties.

Black corals usually do not appear black from the outside. Images: species from different parts of the Indonesian Archipelago, including Antipathes, Stichopathes, Cirrhipathes, Cupressopathes and Rhipidipathes spp., all with different colony morphologies. Photo by Marzia Bo and Tim Wijgerde.
Before Linnaeus, like many other corals, antipatharians were considered marine plants because of their phytomorphic (plant-like) appearance. Therefore, the first illustrations of black corals can be found in old plant catalogues. Black coral spines would still be the most important taxonomic feature if not for a small genus of gorgonians named Dendrobrachia, characterized by a spiny skeleton, which has only recently been moved to the Octocorallia subclass. The principal characteristics distinguishing the order subsequently became the presence of polyps with only six non-retractile tentacles and six primary mesenteries-membranes which extend throughout the polyp coelenteron. Because of their unpinnulated tentacles, black corals were incorporated within the taxon of sea anemones (order Actiniaria). In 1857, Milne-Edwards, on the basis of their colonial habit, considered them sufficiently distinct from actiniarians to create a separated group named “sclerobasic zoantharians”. The detailed microscopic studies of Lacaze-Duthiers, published in 1865, eventually contributed to the recognition of antipatharians as a distinct order of anthozoan hexacorals.
“Black corals have chitinous black skeletons, hence their name.”
The term Antipathidae, used for the first time by Ehrenberg in 1834 for the description of the first antipatharian family, seems to have emerged while describing a family of scleropod bryozoans encrusting a black coral stem. What is certain is that Milne-Edwards and Haime, in 1857, used the same root to define the order name, Antipatharia, and that –pathes became the compulsory and characteristic suffix for all the genera of this taxon.
Systematic literature concerning black corals grew throughout the 19th century until the beginning of the 20th century thanks to great scientific explorations, such as the Siboga Expedition and the Challenger, supplying numerous specimens. After an almost fifty-year gap in taxonomic studies, the finding of new species in the 1970’s started up a major work of taxonomic revision which is still in progress. In addition, the modern explorative campaigns of deep abyssal plains are now shedding light on the enormous biodiversity of these organisms, especially on continental shoals and seamounts. However, the classification of antipatharian corals has been complicated by the existence of species established only on the basis of dry, incomplete museum material, and by the absence of clearly, universally recognised taxonomic characters defining the hierarchy of the order.
Traditionally, the classification of antipatharians has been based on morphological characters. Since 1970, important efforts have been made to enhance the systematic status of these organisms using modern technologies for taxonomic purposes, including molecular analyses. The first molecular work, the complete sequencing of the black coral Chrysopathes formosa genome, was published to support the inclusion of antipatharians in the subclass Hexacorallia, and to separate it from the order Ceriantharia, finally solving a bicentennial debate. A major problem in taxon definition remains the extreme phenotypic plasticity-the ability to change in appearance-of black coral colonies. These differences in colony shape within a given species are caused by variations in environmental conditions, a phenomenon that is also found amongst scleractinian corals. The result is a potential overestimation of the number of species, as different ecotypes-varieties within a species-are confused with true species. It is possible that some micro-features, such as skeletal spines, could be shaped independently from environmental cues. Using scanning electron microscopy (SEM), the study of spine morphology has recently been used with success in taxonomic works, showing microscopic differences in the deposition of skeletal material which are useful for species distinction.
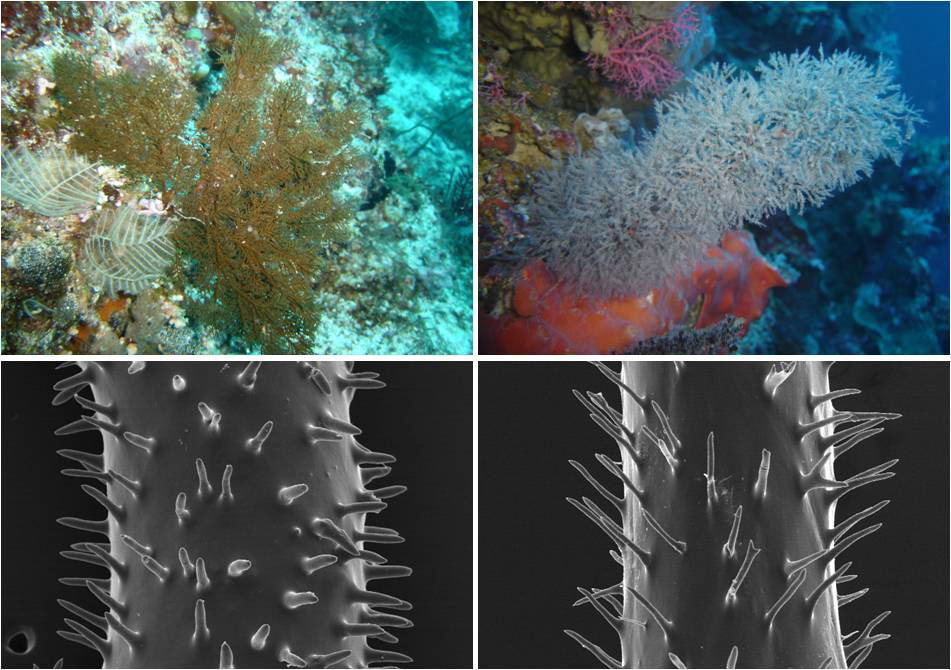
Left column: Myriopathes myriophylla and its spines. Right column: Cupressopathes abies and its spines. Photo by Marzia Bo and Tim Wijgerde.
Nucleotide sequences of coral DNA are an obvious source of additional evidence for systematic relationships of black corals. This approach may be used in combination with morphology, to solve the problem of grouping black corals into the right taxa. The genetic relationships of several species have been studied using internal transcribed spacer (ITS) sequences in black coral DNA, a method frequently used for species identification and evolution studies. Molecular data now suggests that Indonesian species form two major groups: the family Antipathidae and the family Myriopathidae. The degree of genetic variation in these two families differs: in the Myriopathidae cluster, the analysis is not able to separate the species Myriopathes and Cupressopathes in spite of the morphological differences in the colonies, suggesting the possibility of hybridization between these species. In the Antipathidae cluster, the species are more genetically separated. It seems that the degree of genetic variation within the different families reflects the degree of morphological variability of spines. Large differences in spine shape are correlated to a high genetic variability, and vice versa.
Morphology
Black corals greatly vary in shape and size, ranging from a few inches to many feet in height. They are found growing as a single, whirling stem or in a branching pattern. The colony stem is anchored onto the substratum with a basal plate or, in soft bottoms, with a flat hook extremity. The polyps display bilateral symmetry and possess six tentacles, two sagittal and four lateral ones, arranged around an oral cone bearing the mouth at its top. Black coral polyps may show a different set of colours, from transparent to white, yellow, red, orange, pink, green or brown. They have a weak muscular system; their longitudinal muscle fibres are not completely developed and this is the reason why tentacles can only slightly contract. The polyp gastric (coelenteron) is divided by 6-12 mesenteric septa in radial non-communicating chambers, although the coelentera of adjacent polyps are connected.
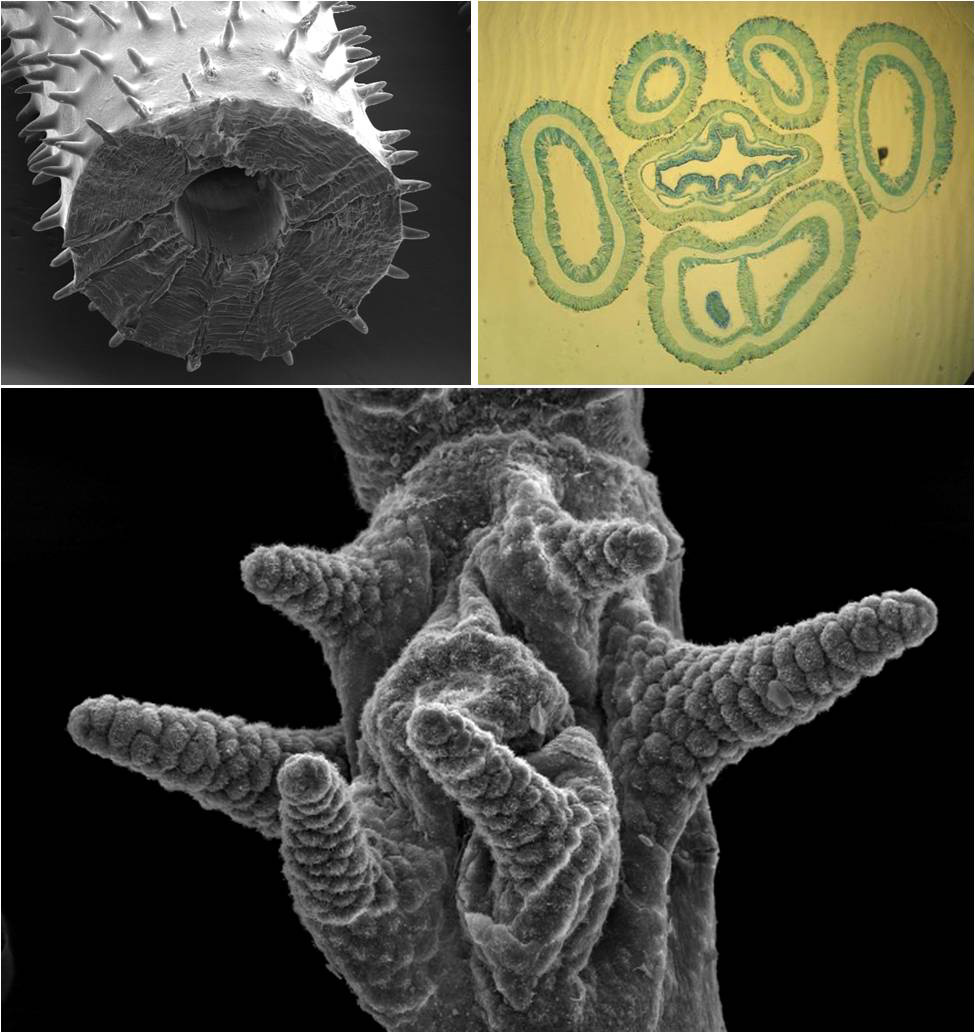
Overview of black coral morphology. Top left: circular, pinnulated skeleton with central canal. Top right: transversal section of a polyp showing mesenteric septa and hollow tentacles. Bottom: Scanning electron microscope (SEM) image of a polyp, showing a typical tentacle arrangement, with four lateral and two sagittal tentacles. Photo by Marzia Bo and Tim Wijgerde.
The skeletons or black coral are circular, with a central canal surrounded by a series of concentric layers of deposited organic material. These microlayers are responsible for the growth rings visible in transversal sections, and can be used to infer colony age and growth rate. The colour of the skeleton varies from black in the thicker portions to golden yellow at the colony tip. All around the branches, the skeleton rises up in a series of small spines arranged in more or less regular longitudinal rows, characterised by distinct features depending on the species. No mineral elements such as spicules or sclerites are present in black coral tissue, which as a result is soft and fragile. The organic material composing the skeleton is produced by the overlying tissue, similar to gorgonians and stony corals, and is known as Antipathin. This substance consists of chitin, a polysaccharide which also makes up the cuticle of insects and crustaceans, and a non-fibrous scleroprotein, which differs from the collagen of gorgonians.
The resistive and elastic properties of the skeleton, which depends on both composition and architecture, are important as these corals encounter significant water flow in their natural environment. For example, net-like Rhipidipathes reticulata colonies living on vertical walls have thin branches which are intricately fused together, providing the skeleton with significant resistive qualities. Antipathes and Cirrhipathes colonies are flexible and resistant, while Stichopathes stems adapted to the strongest currents are thicker and more rigid, but commonly go through skeletal fractures.
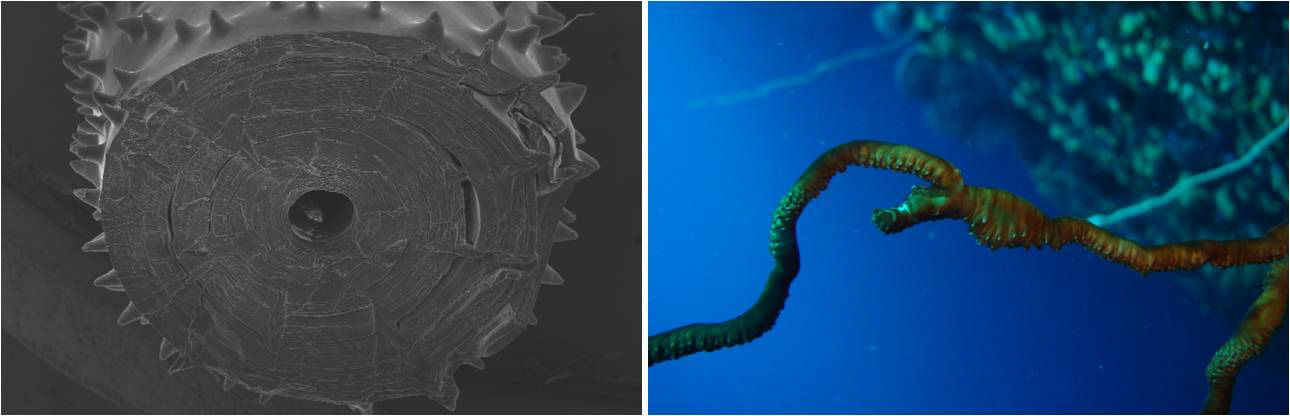
Left: growth rings making up the circular skeleton. Right: anomalous growth of a Stichopathes whip coral after multiple fracture events. Photo by Marzia Bo and Tim Wijgerde.
It is interesting to note that unlike gorgonians, where collagen fibres determine flexibility, in black corals chitin is responsible for the elastic properties while polyphenolic proteins contribute to skeletal reinforcement. Spines play an important role in the protection of polyps. Recent studies highlight the role of spines as points of discontinuity in chitin fibre arrangements, enhancing the resistance of the skeleton against torsion created by water current.
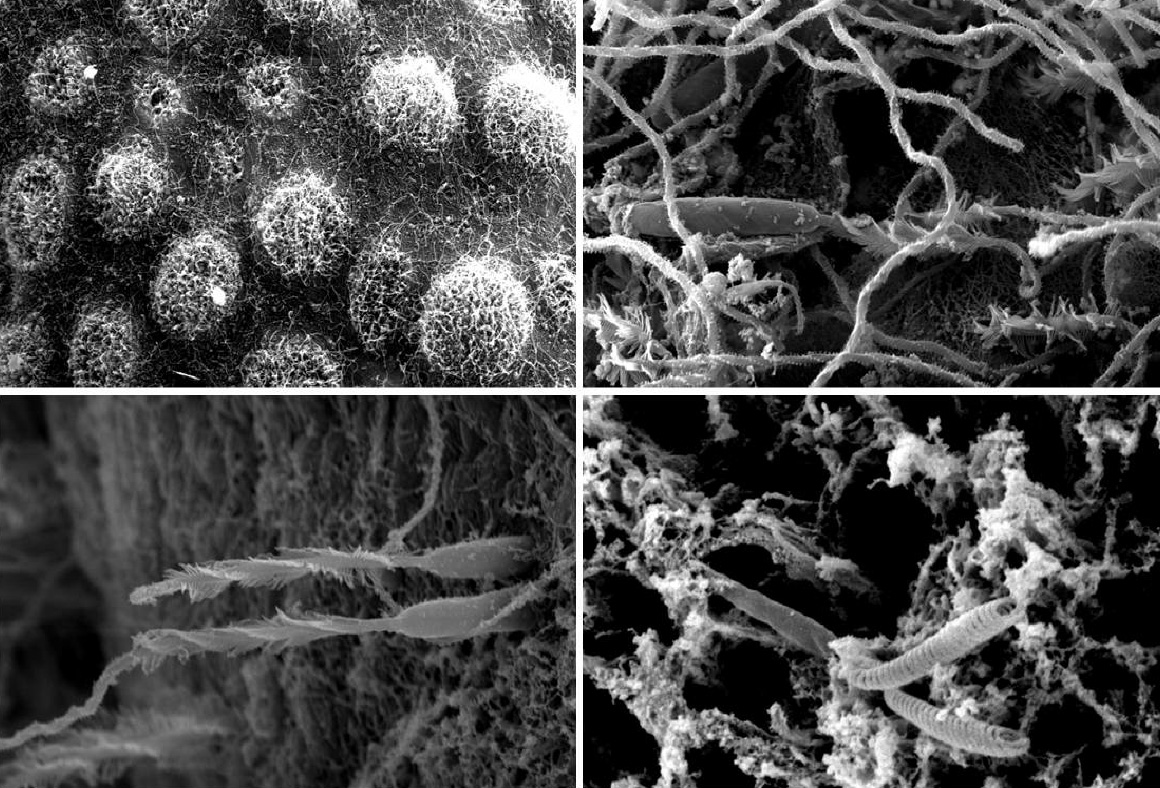
Examples of black coral cnidocytes. Nematosores, clusters of cnidocytes, are visible on tentacle surfaces, including various types of cnidocysts such as basitrich isorhizas, mastigophores and spirocysts. Photo by Marzia Bo and Tim Wijgerde.
Like all cnidarians, black corals have stinging cells, or cnidocytes. Their complete set, also known as the cnidome, typically consists of spirocysts, microbasic mastigophores, and basitrich and holotrichous isorhizas. These various cell types have roles in colony defense and prey capture, and are arranged differently depending on species. Field observations on whip black corals show that their large polyps can extend their tentacles up to ten times their normal length. These so-called sweeper tentacles respond to disturbing corals or encrusting animals, firing their cnidocytes in defense. Not all organisms suffer from this attack; spongesm for example show a strong anti-fouling reaction, rapidly destroying black coral tissue.
Reproduction

Left column: sweeper tentacles produced after physical contact with disturbing organisms. Right column: disturbance of antipatharian tissues by gorgonians and soft corals. Note the exposed black skeleton in the lower right image. Photo by Marzia Bo and Tim Wijgerde.
Black corals are known as gonochoric organisms, meaning that separate male and female colonies are found. Generally, the entire colony has the same sex, although hermaphroditic species have been found. Of the six primary mesenteries, only the two placed in the sagittal direction bear reproductive organs, and they are usually the longest. Observations conducted in the field have always reported the absence of clear external morphological differences between the sexes. This seems to be the general rule, both in fertile and infertile colonies.
“Anchorages of broken fragments are not regenerated, suggesting that fragmentation is not a common reproductive strategy for black corals.”
Some species, namely Antipathella subpinnata and Antipathella fiordensis, seem to be exceptions. In fact, in both species the pink fertile polyps form a clear contrast with the non-fertile white zooids. Fertile polyps are distributed in random patches in branched antipatharians, while in whip black corals the apical portion is generally always fertile in mature colonies, and fertility along the rest of the stem is more random. The quantity of eggs per polyp generally depends on its size. For example, large female polyps of Cirrhipathes spp. (2-3 mm in transverse diameter) can carry up to five hundred eggs each and show continuous phases of fertility throughout the entire year.
Recent microscopic evidence of Indonesian specimens indicates that sperm cells originate from flagellated gastrodermal cells, and are successively expelled from the mouth. Pro-acrosomal vesicles and a 9+2 flagellum are considered to be a feature of primitive spermatozoa, which are adapted to external fertilization. This is in accordance with the general idea of antipatharians as being one of the most primitive groups of anthozoans. Another interesting feature is a cup-like body, found around the flagellum, whose function is probably related to tail movement, and whose occurrence seems to be limited to the sperm of antipatharians.
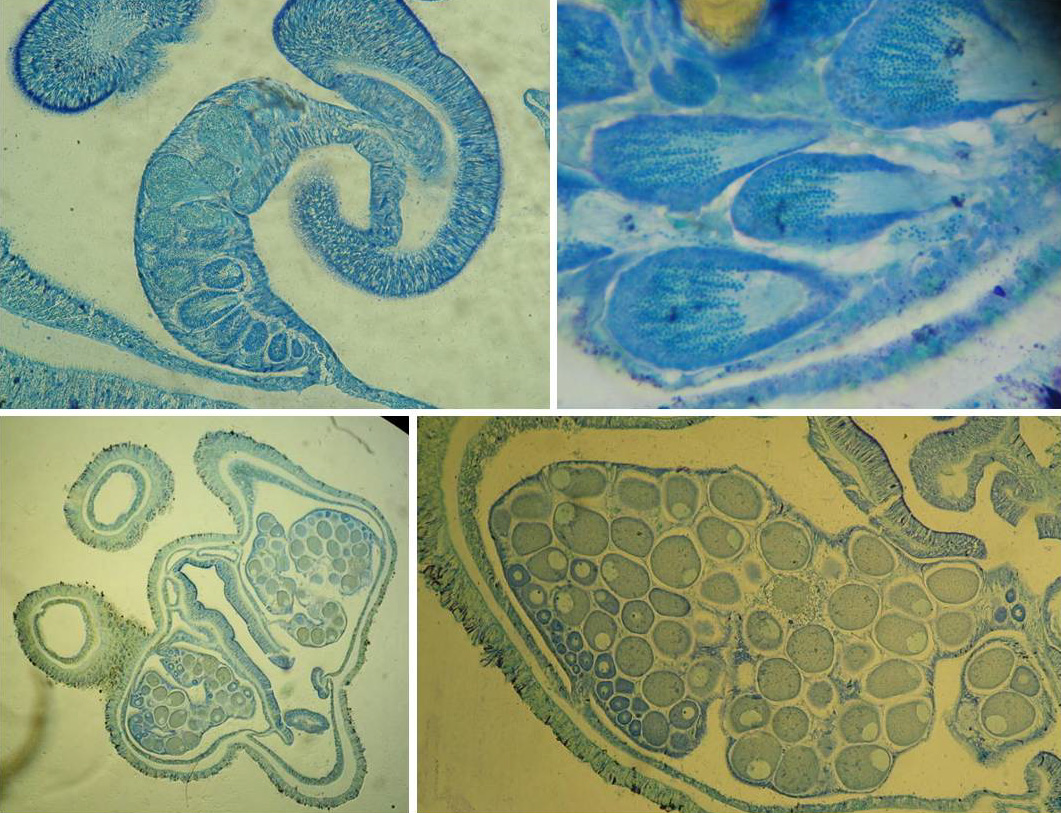
Reproductive organs of black corals. Top: sperm cysts in reproductive mesentery. Bottom: eggs in reproductive mesenteries. Photo by Marzia Bo and Tim Wijgerde.
The importance of asexual processes has been verified through long-term monitoring of Indonesian whip black coral meadows, where signs of apical (top) fractures have been reported for the majority of colonies. These are likely related to water current. It seems that these species undergo frequent episodes of fracture and regrowth. Anchorages of broken fragments are not regenerated, suggesting that fragmentation is not a common reproductive strategy for black corals. Either way, asexual reproduction plays an important role in the reproduction of black corals. Budding is the most common strategy, and this is the reason why alternation of adult and juvenile polyps on branches is observed frequently.
Larvae have been described only for Antipathella fiordensis as negatively buoyant, non-feeding and short-lived planulae (approximately 10 days). These factors, coupled with observations of restricted water movement in the fjords where this species lives, suggest that larval dispersal and gene flow are limited and that separate fjords may represent genetically isolated populations. Considering the habitat preferences of adults, it has been hypothesized that they tend to settle on inclined substrata, with low light intensity and high nutrient supply. Laboratory observations conducted on cut tentacles of Indonesian black coral polyps have revealed their high resilience (kept alive in cups for many days) and their ability to reorganise into independent structures. Active ciliate motion on their surface makes them move around in a peculiar fashion. It is possible that these observations may represent pseudo-larvae, representing the phenomenon of “polyp bail-out”, also described for A. fiordensis.
Ecology
A few years ago, a rich antipatharian community in the Marine Park of Bunaken (North Sulawesi, Indonesia) was discovered, counting around forty species. This discovery allowed new research to be undertaken, shedding new light on several biological and ecological aspects of black corals, such as species richness, habitat preferences, specific depth ranges, symbiotic associations, feeding behaviours, reproductive strategies, growth rates and phylogenetic relationships. Black corals seem to be keystone species, essential for the maintenance of high biodiversity levels on coral reefs.
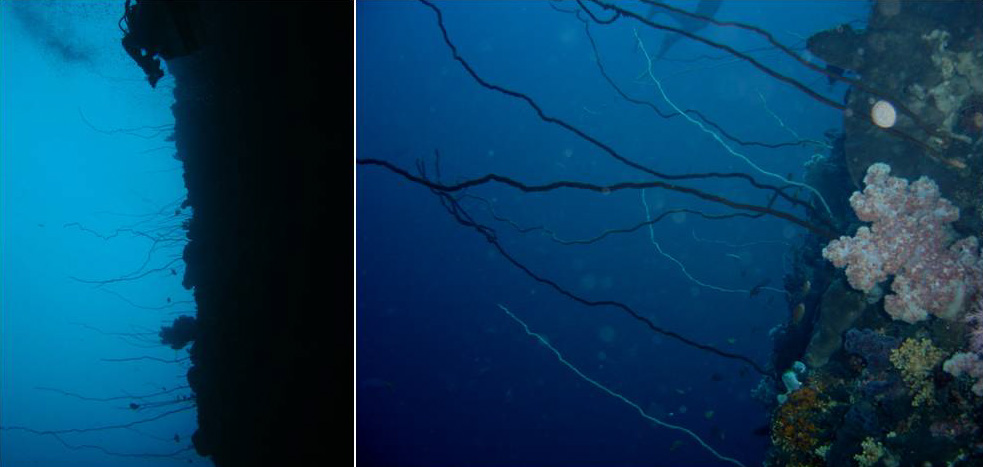
Meadows of whip black corals belonging to the genera Stichopathes and Cirrhipathes. Photo by Marzia Bo and Tim Wijgerde.
The bathymetric spectrum, or depth range of black corals, is extremely wide. Members of four of the seven antipatharian families (Schizopathidae, Leiopathidae, Stylopathidae and Cladopathidae) are almost exclusively deep species. This is especially true for the Schizopathidae family, whose species show strong adaptations to soft bottoms between 200 and 2,000 m in depth, such as hook-like anchorages. In contrast, the Antipathidae, Aphanipathidae and Myriopathidae families contain more species with shallower habitats. For example, the genus Cirrhipathes is found mainly in the coastal zone, being a so-called stenobath genus as it occurs within a limited depth range. The minimum depth limit of this genus is determined by low tide level, as they generally do not tolerate prolonged air exposure caused by tidal movements. Other genera are eurybath, meaning they are found in wider depth ranges depending on location, including Antipathes and Bathypathes.
Some areas are particularly suitable for massive colonization by deep water black corals. Examples are the New Zealand southern fjords and the Colombian coastal zone, which are characterized by cold waters, high nutrient levels due to upwelling or land run-off, low light intensity, little water turbulence and limited competition. Thus, black corals can thrive in areas where many other corals cannot.
In Bunaken, the number of black coral specimens increases with depth, reaching maxima at 117-150 feet (35-45 m). This includes several typical deep water corals, such as Cupressopathes and Rhipidipathes spp. Their depth distributions and habitat preferences are shaped by biotic and abiotic factors. The most likely biotic factor is competition for space with stony corals in the shallow area of the reef. This fierce competition is probably why black corals show increasing abundance and diversity with depth, as also observed on Caribbean reefs. The only way an antipatharian can compete at low depths is to settle in crevices amongst stony corals, and in fact, only the unbranched Cirrhipathes contorta coexists with scleractinian corals. This species occupies a minimal amount of substrate surface and is able to adapt its shape to the available space.
Water current, in terms of intensity, pattern and direction, is a major abiotic factor. Similar to other filtering colonial organisms, black corals tend to settle in areas of moderate to strong current, maximizing food capture and larval dispersion. Indonesian coral reefs can host huge meadows of whip corals at sites with receive sufficient water flow. Water movement acts not only on the distribution of species, but also on their morphologies. Colony morphology is a function of age and water current. A colony may show strong phenotypic plasticity depending on the intensity of the current: strong currents lead to more numerous and thicker branches, to maximize filtration efficiency and friction resistance. Where no prevailing flow direction is detected, colonies display a three-dimensional bushy morphology. These field observations have implications for their husbandry requirements (see below). In Bunaken, black corals are adapted to moderate and strong currents. Branched corals may live exposed on vertical reefs as fan-shaped colonies, or in canyons as arborescent colonies receiving vertical currents. Some species occupy the roofs of narrow gorges and caves, scattered on the reef wall. These species have thin, delicate, bushy colonies, which avoid sedimentation on the bottom of their hole and entrap everything rolling down from the reef, both living epibionts and detritus.
Feeding strategies

An Antipathes specimen entrapping a great amount of coralline algae and other organisms with its branches. Photo by Marzia Bo and Tim Wijgerde.
Next to affecting colony morphology, water current has a major effect on coral feeding rates. Being suspension feeders, black corals tend to orient themselves in the best way possible to the water current to maximize food capture. For example, whip corals such as Stichopathes spp. are adapted to strong currents, and they do not orient their fragile polyps directly towards the current. Lacking mineral sclerites, their tissues cannot withstand strong shear forces. Instead, their long sagittal tentacles extend perpendicularly to the lateral ones, capture prey and redirect them to the mouth. In black coral meadows, the majority of colonies arranges its polyps in the same way. Cirrhipathes spp. have polyps arranged all around the stem, being able to capture food particles from every direction. Species with bottlebrush morphologies such as Cupressopathes spp. utilize the same strategy, representing a compromise between a highly branched colony and minimal frictional stress.

Top left and right: specimen of Stichopathes sp. with expanded polyps during nocturnal feeding activity and contracted tentacles during the day. Bottom left: expanded basket-like polyps of the whip coral Cirrhipathes anguina. Bottom right: net-like branching pattern of Rhipidipathes reticulata. Photo by Marzia Bo and Tim Wijgerde.
Some species are able to completely contract their tentacles, such as Cirrhipathes and Stichopathes, whilst other never contract them, such as Rhipidipathes. Yet other species only partially contract their tentacles. The observed circadian rhythms of expansion and contraction have been correlated to the energetic balances of various species. Large basket-like polyps such as those of Cirrhipathes only expand at night to reduce energetic costs, and are typical for whip corals adapted to strong currents. They direct their long tentacles upwards to capture large zooplankton. In contrast, small net-like polyps such as those of Rhipidipathes are always expanded, and are typical for fan-shaped corals adapted to moderate currents. Their tentacles are directed laterally, and they almost touch one another to increase filtration efficiency.
Laboratory experiments on Antipathes grandis reveal that its polyps are able to capture amphipods, copepods and chaetognaths (arrow worms). Tentacles capture prey, after which microscopic cilia on the ectodermal cells move prey along a mucus film towards the mouth. In Indonesia, the production of such films during feeding has never been observed. Instead, mucus is always produced copiously after touching the colony, as a defensive response.
“Large basket-like polyps only expand at night to reduce energetic costs. Small net-like polyps are always expanded, and are typical for fan-shaped corals adapted to moderate currents.”
It has also been reported that several polyps may cooperate in the capture of large prey. A gut content analysis of basket-like polyps, such as those of Indonesian Cirrhipathes spp., demonstrated that large zooids show a predatory activity on small zooplankton. Moreover, small groups of 2-3 polyps may also collaborate to trap large polychaetes. Next to suspension feeding using tentacles, mucus nets and discharge of spirocyst microfibrils, black corals also use filter feeding. By slowly drawing in water with cilia, polyps ingest unicellular and particulate food material. This is especially true for species with small, net-like polyps, which prefer nano- and microplankton (2-200 µm size range, including algae, protists and large bacteria) and particulate organic matter, entrapped with mucous nets.
Symbiosis
Since in the early literature of black corals, colonies have been reported as a true oasis for organisms, whether sessile or mobile. The most commonly observed epibionts are commensal invertebrates: cirripeds (barnacles) are frequent inhabitants of branched antipatharians, in particular lepads of the genus Oxynaspis. The spiny skeleton of the corals typically covers these animals and at times may also generate small branches. These symbionts are also found in scleractinian corals. SEM photographs distinctly show the coverage, as well as anomalous deposition of skeletal spines. Interestingly, spines don not grow all over the lepad shell. They tend to leave the opening for the crustacean’s cirri free, allowing them to come out. In Indonesian samples, gastropods are also frequently found, covered by black coral skeleton. Polychaetes are also common symbionts. The worm Marphysa antipathum is able to build parchment-like tubes armed with sponge spicules and is a typical black coral epibiont. Branched antipatharians are very suitable habitats and in some cases, the tube-like arrangement of pinnules is used as permanent refuge. It has also been hypothesized that the commensal worm may stimulate and direct the growth of these galleries. Next to cirripeds and polychaetes, many crustacean decapods such as crab and shrimp species have been described as species-specific epibionts of tropical black corals, showing clear mimicries. Examples are Periclimenes wirtzi and Pontonides spp. It seems that while crustaceans benefit from a protected habitat and food collected by polyps, the corals gain no advantage. In some cases, when food is scarce, crabs may even start feeding on coral tissue, similar to Trapezia crabs in stony corals. The presence of ophiuroids (brittle stars) has also been frequently reported in the literature. Antipathella fiordensis hosts the obligate brittle star Astrobrachion constrictum. This epibiont feeds on polyp mucus during the night, while it exploits captured prey during the day. In return, the feeding movements of the ophiuroid arms clear the coral colony of suspended material that might otherwise smother the polyps. Field observations following natural catastrophic events and transplant experiments suggest that survival of A. fiordensis is higher when the brittle star is present. Other epibionts reported in the literature are sponges, anemones (e.g. Nemanthus sp.), bryozoans, crinoids, caprellids (skeleton shrimp), serpulids and spirorbids (calcifying tube worms), parasitic ovulid snails (Phenacovolva spp.), other corals, small oysters and even fishes. An interesting example of fishes are certain Bryaninops gobies, whose reproductive cycle is totally dependent on the existence of the coral.
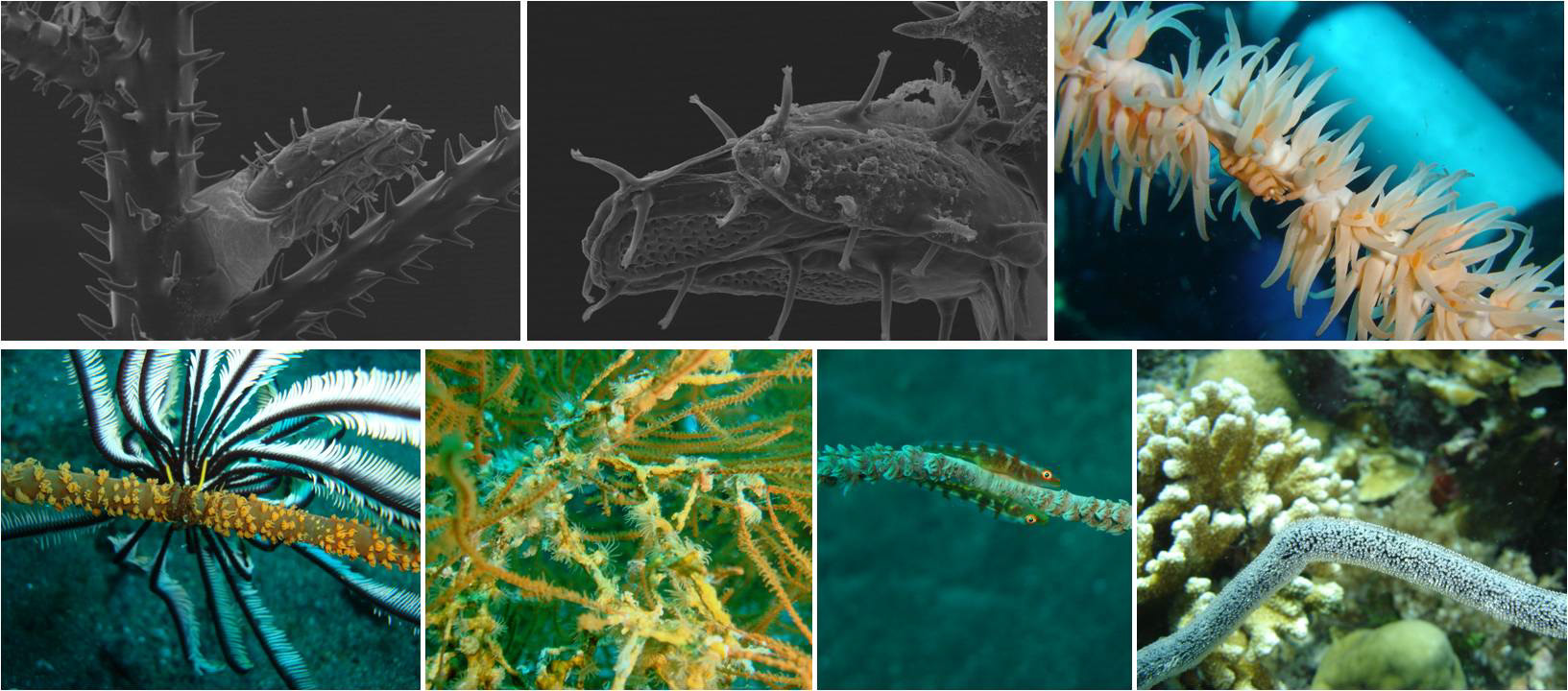
Overview of several black coral epibionts. Top and middle left: lepads covered by the black coral’s spiny skeleton. Top right: mimic Pontonides shrimp living on a colony of Pseudocirrhipathes mapia. Bottom left: a crinoid holding on to a coral stem. Bottom middle: parasitic zoanthids on an Antipathes colony, gobiid fish living on Cirrhipathes anguina. Bottom right: fish eggs laid on a naked antipatharian skeleton. Photo by Marzia Bo and Tim Wijgerde.
In Bunaken, black corals also host a highly diversified epibiotic fauna, which probably finds shelter due to the three-dimensional arborescent shape of the colonies. Generally, the diversity of the epibiotic community is related to colony shape. On unbranched colonies, usually only one epibiont type exists, often a couple of crabs or shrimp. On fan-shaped and arborescent colonies, a wider range of species may be found.
Together with the epibiosis of living colonies, dead black coral skeletons are very common secondary substrates for colonisation. Recently, a conservative estimate was made of the total number of living organisms occupying a single dead colony of Antipathes dendrochristos, which was about 2,500 individuals! It is clear that while some groups of invertebrates are found in high densities on both live and dead colonies, others appear to be mostly limited to either living or dead colonies. A similar phenomenon is described for the Mediterranean black coral Antipathella subpinnata, found in the Strait of Messina, which are impacted by abandoned fishing lines scratching coral tissue and enhancing the colonisation of numerous encrusting epibionts.

Many black corals have recently been found to host zooxanthellae (image: an Antipathes colony at 100 feet, Mentawai Islands, Indonesia). Photo by Marzia Bo and Tim Wijgerde.
Until recently, black corals were thought to be unique hexacorals due to the complete absence of zooxanthellate species. In the 20th century, only one author reported the presence of few symbiotic zooxanthellae in the endoderm of some Cirrhipathes species. However, the scarcity of information made this finding doubtful and it has never been sufficient for researchers to consider the order zooxanthellate. In 2011, however, two reports of zooxanthellate black corals from Hawaii and Indonesia were published. Cirrhipathes specimens from Indonesia can host approximately ten million zooxanthellae per mm2 in their gastroderm. Molecular analysis has revealed these dinoflagellates to be closely related to the symbionts of clionid sponges (Symbiodinium clade G). There was also evidence for additional symbionts in clade C, at low abundance. The high abundance, the presence of symbiosomes (intracellular vesicles which contain the zooxanthellae) and reproduction of the zooxanthellae in the coral all suggest that they provide the coral with nourishment. This, again, has important implications for the husbandry of these corals (see below). What is most striking is that Hawaiian zooxanthellate corals-Stichopathes, Acanthopathes, Bathypathes and possibly Myriopathes spp.-were found up to a depth of 1,320 feet (396 m). The authors theorize that in many cases, the corals occur too deep (below 417 feet or 125 m) for their zooxanthellae to carry out photosynthesis and be beneficial. In deep waters, these symbionts may act as parasites (drawing nutrients from the coral) or commensals (having no effect on the coral) rather than mutualists. They also suggest that the energy demand of these dinoflagellates could either be reduced by dormancy, met by self-digestion, or by feeding on an external food source.
Growth and Transplantation
Relatively few studies have taken the growth strategies of black corals into consideration. The majority of authors have focused on the chemical composition, structure and architectural mechanics of black coral skeleton. Black corals are generally considered slow-growing organisms, with long life spans and older age of maturity. Literature data concerning growth rates focus mainly on radial growth rates recorded with radiometric studies using radioactive carbon (14C). Isotopic studies on organic and carbonate coral skeletons in fact allow for palaeoclimatic reconstructions of superficial and deep water temperatures in the recent past. Concerning growth in height, studies have focused on branched antipatharians of tropical and temperate waters, revealing low rates ranging between 0.5 cm and 9.3 cm per year. However, unbranched black corals such as Cirrhipathes spp. can grow at very high rates of 13.25 cm per month, which is among the highest rates ever recorded for colonial cnidarians.

Transplanted fragments of Stichopathes sp. showing growing extremities. Photo by Marzia Bo and Tim Wijgerde.
In order to start restoration projects of damaged reefs, it is key to assess the feasibility of black coral transplantation. Information on growth rates, longevity and reproductive cycles of black corals is vital to conservation efforts. In all transplantation experiments reported in the literature, critical factors proved to be chosen habitat, light availability, current intensity, and substratum. Long-term experiments conducted on whip black corals in Indonesia highlight the role of these species as targets for transplantation. Highest survival rates are displayed by adult colonies, which can be fragmented easily. In this case the apex will show the fastest growth rate, which is especially true for Cirrhipathes cf. anguina. This species clearly shows a strong phototropic (light-stimulated) growth that orients upward. Colonies which settle on substrata of different inclinations have confirmed the important role of light in black coral development.
Aquaculture and Husbandry
The over-exploitation of shallow water black corals for commercial purposes, mainly due to the jewellery trade, has led to the inclusion of the entire Antipatharia order in the IUCN list and in Appendix 2 of CITES. The most endangered reefs in this respect are those of Hawai’i, Tonga, Ecuador and numerous Caribbean localities. Indonesian reefs are under less pressure caused by the black coral trade. The recent scientific insights described above give aquarists the opportunity to explore the possibilities of sustainable aquaculture. Together with mariculture, this could provide a steady flow of eco-friendly fragments. Unbranched Stichopathes and Cirrhipathes ssp. from the Antipathidae family seem to be key candidates for asexual propagation, as they have high growth rates. It is however unclear whether survival rates of artificial fragments are high enough to make this approach viable.
Another issue are the husbandry requirements for black corals in general. From field studies, it is clear that water flow, nutrition and light are important factors. First of all, a moderate water flow rate of 2-4 inches per second (5-10 cm s-1) may be appropriate for Cirrhipathes spp. Higher flow rates above 4 inches s-1 may be suitable for Stichopathes spp., which are adapted to strong currents, although no data exist on prey capture rates in relation to flow rate. As black corals usually grow below 33 feet (10 m), water flow probably needs to be quite laminar as well, and either uni- or bidirectional. Bidirectional currents, changing every e.g. six hours to simulate tidal movements, may be suitable for shallower species which could be exposed to the tides. Cirrhipathes spp. have polyps arranged all around their stems, being able to capture food particles from every direction, suggesting that changing flow directions may benefit species from this genus. Either way, laminar flow patterns are hard to achieve in the average aquarium, as the straight angles of the aquarium, together with live rock, create significant turbulence. A raceway-style aquarium may therefore be more appropriate. In addition, precise measurements of water flow rate require equipment not available to the average aquarist.

A Stichopathes colony in an aquarium, growing in a typical spiral-shaped pattern. Photo by Marzia Bo and Tim Wijgerde.
The light-oriented growth of Cirrhipathes spp., together with the zooxanthellate nature of many black corals, suggest that corals collected from shallow waters should not be kept in constant darkness. This begs the question of what irradiance levels are appropriate. Given the depth range of collected shallow antipatharians, likely around 33-100 feet (10-30 m), they can experience light intensities that are considered moderate to very high to aquarists (between 200-1,000 µmol quanta m-2 s-1). The spectral quality of this light will also be skewed towards the blue part of the spectrum. This does not necessarily mean black corals require high irradiance in the aquarium, as it is still unclear to what extent these corals benefit from photosynthesis.
Finally, next to water flow and light, nutrition is important. As black corals may rely heavily on heterotrophic feeding, that is the capture, digestion and assimilation of organic substances, they require regular feedings when kept in aquaria. As many black corals are known to feed on zooplankton and detritus on reefs, copepods, Artemia nauplii, rotifers (Brachionus) and dry feeds seem suitable food items. It is recommended to feed these corals small batches every couple of hours, by using feeding timers or peristaltic pumps. Nano- and microplankton (algae, protists and large bacteria) are also prime candidates for feed testing. Daily additions of phytofeast, containing a cocktail of green algae, diatoms, haptophytes and bacteria, may be especially beneficial to species with small, net-like polyps, such as Rhipidipathes spp. If black corals are provided with sufficient food items, they may rely less heavily on light and the photosynthetic products provided by their zooxanthellae. Future aquarium studies will have to address the issue of heterotrophy versus photoautotrophy in black corals.
Another matter, linked to food availability, is the filtration system used. Whatever means of filtration used, it is key that sufficient food particles remain in the aquarium water throughout the day and night. Recently, an article about Dynamic Mineral Control (DyMiCo LINK) was published in this magazine, which is a filtration system that maintains live plankton in the water whilst removing inorganic nutrients. A hobby-scale version of this system is now in development, and at the time of writing, the first black corals (Cirrhipathes sp.) are being tested.
In Conclusion
It should be clear by now that antipatharians are fascinating corals that deserve our attention. Next to their ecological and commercial value, the organic skeleton of black corals may have biomedical applications. By making use of sustainable mariculture and aquaculture only, the aquarium industry can play an important role in the conservation of black corals. It is therefore vital that we continue to expand our knowledge on the biology and husbandry requirements of these corals.
References
- D. Wagner, X. Pochon, L. Irwin, R. J. Toonen and R. D. Gates. 2011. Azooxanthellate? Most Hawaiian black corals contain Symbiodinium. Proceedings of the Royal Society B 278: 1323-1328
- E. Gaino, M. Bo, M. Boyer and F. Scoccia. 2008. Sperm morphology in the black coral Cirrhipathes sp. (Anthozoa, Antipatharia). Invertebrate Biology 127: 249-258
- H. F. N. Lapian, M. Barucca, G. Bavestrello, M. A. Biscotti, M. Bo, A. Canapa, S. Tazioli, E. Olmo. 2007. A systematic study of some Black Corals species (Antipatharia, Hexacorallia) based on rDNA internal transcribed spacers sequences. Marine Biology 151: 785-792
- K. R. Grange. 1991. Mutualism between the antipatharian Antipathes fiordensis and the ophiuroid Astrobrachion constrictum in New Zealand fjords. Coelenterate Biology: Recent Research on Cnidaria and Ctenophora Developments in Hydrobiology 66: 297-303 Onderkant formulier
- M. Bo, A. C. Baker, E. Gaino, H.H. Wirshing, F. Scoccia and G. Bavestrello. 2011. First description of algal mutualistic endosymbiosis in a black coral (Anthozoa: Antipatharia). Marine Ecology Progress Series 435: 1-11
- M. Bo, C. Di Camillo, A. M. Addamo, L. Valisano and G. Bavestrello. 2008. Growth strategies of whip black corals (Cnidaria: Antipatharia) in the Bunaken Marine Park (Celebes Sea, Indonesia). Marine Biodiversity Records 2: 1-6
- M. Bo, G. Bavestrello, S. Canese, M. Giusti, E. Salvati, M. Angiolillo and S. Greco. Characteristics of a black coral meadow in the twilight zone of the central Mediterranean Sea. Marine Ecology Progress Series 397: 53-61
- M. Bo, M. Barucca, M. A. Biscotti, A. Canapa, H. F. N. Lapian, E. Olmo and G. Bavestrello. Description of Pseudocirrhipathes (Cnidaria: Anthozoa: Hexacorallia: Antipathidae) a new genus of whip black corals from Celebes Sea. Italian Journal of Zoology 76: 392-402
- M. Bo, S. Tazioli, N. Spanò and G. Bavestrello. 2008. Antipathella subpinnata (Antipatharia, Myriopathidae) in Italian seas. Italian Journal of Zoology 75: 185-195
- S. Tazioli, M. Bo, M. Boyer, H. Rotinsulu and G. Bavestrello. 2007. Ecology of some common antipatharians from the Marine Park of Bunaken (North Sulawesi, Indonesia). Zoological Studies 46: 227-241







Very nice collection with brief explanation.