Background
In March, 2003, Dr. Ronald Shimek published the results of a bio-assay study in which he concluded that two of the most popular salt mixes in the aquarium industry, Instant Ocean and Coralife, were toxic to sea life (1). The study further concluded that two other less common salt mixes, Bio-Sea MarineMix and Crystal Sea Bio-Assay Formula, fared significantly better – comparable to natural sea water (2). Dr. Shimek indicated that the most likely cause of this toxicity was the level of heavy metals present in the salt mixes (3).
Like most aquarists, we were immediately concerned over these results. As the title of Dr. Shimek’s work states, toxic metals in our salt mixes are indeed a bad beginning for a reef aquarium. Further inspection of the study revealed several areas in which further testing and study might be of value.
First, the data on heavy metal concentrations for these four salt mixes had come from two very different sources: Instant Ocean and Coralife from a four year old independent analysis (Atkinson & Bingman, 1999) and the other two from the manufacturer. Dr. Shimek does express some concern over this (4), but concludes that the manufacturer’s data is not suspect (5). This was an interesting conclusion in light of the work that Dr. Shimek and we had done on Two Little Fishies Combisan two years prior (Inland Reef and Ron Shimek 2001). In that case, the data printed on the bottle, from a very well-respected manufacturer, was found by Dr. Shimek to be completely unreliable. In the controversy that followed, Inland Reef conducted an independent analysis andconfirmed Dr. Shimek’s data, eventually resulting in the manufacturer removing the printed analysis from their bottle.
Second, if obviously high toxic metals levels in Instant Ocean and Coralife were the root cause of the poor bio-assay performance in Dr. Shimek’s study, we reasoned that conducting an analysis on all four salts, handled and testing in an identical manner, might reveal the causative element or elements.
Finally, we had it good authority that the chemical formulas for many salt mixes had been updated since Atkinson and Bingman’s 1999 study. This valuable data was badly in need of being updated using today’s salt mix formulations.
We therefore resolved to conduct an independent elemental analysis of the salt mixes. After discussions on Reefs.org, the scope of the work grew to cover all of the popular salt mixes currently used in the hobby, not just those in Dr. Shimek’s study. The high cost of performing so many elemental analyses led Inland Reef to seek donations to assist with the total costs. The vast majority of funding for this study was provided by members of Reefs.org and several auctions of donated merchandise conducted by Reefs.org. There were also contributions from the members of ReefCentral.com and a matching donation from the ReefCentral.com staff. The remainder of the cost was covered by Inland Reef Aquaria directly.
Unfortunately, we made the decision to close Inland Reef Aquaria and pursue other business interests just as the study was getting underway. This and several other “real-world” responsibilities led to repeated delays in the study and the publication of its results. We apologize sincerely to all of you who have contributed to this effort and have patiently awaited the results for far longer than anyone ever anticipated. We hope you will find it worth the wait!
Procedure
The selection of salts to be tested was based on a series of polls conducted on the Reefs.org discussion forum. Only a limited number of tests could be performed with the available funds. In addition, poll results favored the inclusion of Oceanic’s new salt mix over Coralife, causing one of the salts in Dr. Shimek’s study to be dropped from the list. The final list, in no particular order, is:
- AquaMedic Sea Salt (AM)
- Oceanic Sea Salt (O)
- Omega Sea Marine Salt (OS)
- Aqua Craft Bio-Sea MarineMix (BSM)
- Marine Enterprises Crystal Sea MarineMix (CSM)
- Tropic Marin Sea Salt (TM)
- Instant Ocean (IO)
- Kent Marine Salt (KM)
- Marine Enterprises Crystal Sea Bio-Assay Formula (CSB)
The abbreviations indicated here are used on the graphs and tables to be found below.
Several bags (or boxes) of each salt were obtained over a 10 month period. All salts were either purchased from wholesale suppliers or retail stores, or obtained directly from the manufacturer at trade shows. In no case was any manufacturer aware that they were providing a sample to be used in this study.
Several samples were obtained from each bag, from various positions within the bag, to minimize the effects of settling. In a perfect world, several elemental analyses would be performed on each salt brand, allowing an error range to be determined for each element tested. Unfortunately, the high cost of these analyses ruled out performing that many tests – an initial estimate to perform three tests per salt mix came to over $15,000. Therefore, the samples from at least three different bags of the same brand salt were combined to obtain an “average” sample for each brand. We then made sure that the testing lab performed each batch in succession with
calibration runs interleaved. We also included a reference sample of natural sea water and RO/DI water with each run. The raw data for the NSW and RO/DI samples gave us a very high confidence in the data for all elements tested. In addition, we are reasonably certain that the period over which we obtained the sample bags did not include formula changes by any manufacturer.
Once the samples were taken, each was assigned a random code letter. All subsequent handling and testing was performed “in the blind” by someone who did not know the code letter assignments. Only after the results came back from the lab were they re-associated with the actual salt mix names.
All labware that came into contact with the salts, including mixing and measuring containers, sample containers, stirring rods, sampling spoons, etc., were triple acid-washed polypropylene to minimize the possibility of any metals contamination. Separate sets of labware were used for each salt mix to prevent any cross-contamination.
The salts were mixed with RO/DI water to a concentration of 35 ppt, as measured with a calibrated refractometer. A sample of the RO/DI water was included in each batch of samples sent to the lab. The RO/DI water tested below detection limits on all elements except Zinc, which measured 21 ppb. This value was subtracted from the Zinc levels reported for each salt sample.
Yield Test
To test yield, we measured the dry weight of each salt mix required to produce a given amount of salt water at a concentration of 35 ppt. To keep moisture accumulation to a minimum, the length of time each dry sample was exposed to the air was minimized and all dry samples were handled in an identical manner. After all the salt solutions were mixed, the salinity was tested with a calibrated refractometer. The wet samples were then allowed 24 hours to fully dissolve before re-testing and, if needed, adjusting the salinity to 35 ppt. The table below shows the amount of dry sample used, the volume of salt water produced, and the actual yield from a 50 gallon bag (or equivalent portion) of each salt:
| Salt Mix | Dry Weight (g) | Yield (ml) | ml produced per g | 50 gal bag yields |
|---|---|---|---|---|
| Aqua Medic | 21.14 | 536 | 25.355 | 42.26 gal |
| Oceanic | 21.35 | 526 | 24.637 | 42.96 gal |
| Omega Sea | 23.47 | 571 | 24.329 | 43.70 gal |
| Bio-Sea Marinemix | 22.59 | 580 | 25.675 | 46.12 gal |
| Crystal Sea Marinemix | 21.39 | 534 | 24.965 | 43.53 gal |
| Tropic Marin | 21.19 | 540 | 25.484 | 41.74 gal |
| Instant Ocean | 20.70 | 520 | 25.121 | 45.13 gal |
| Kent | 20.67 | 520 | 25.157 | 42.20 gal |
| Crystal Sea Bio-Assay | 20.65 | 510 | 24.697 | 43.00 gal |
There is a difference of 4.38 gallons between the highest and lowest yields (just under a 9% variation). But, clearly, every salt manufacturer plans for a salinity less than full strength sea water when advertising the yield of their bags.
Elemental Testing
Initially, we wanted to perform some basic testing (pH, Ammonia, Nitrite, and Nitrate) before sending the wet samples to the lab since the Inductively Coupled Plasma Mass Spectrogram (ICPMS) testing only gives elemental concentrations. In preparation, we did some preliminary research on consumer test kits, as well as performing some basic tests on reference samples. We came to the unfortunate conclusion that no consumer test kit on the market today is accurate enough for these purposes. In some cases, even a basic trend was difficult to obtain (i.e., higher concentration reference samples sometimes failed to actually test higher). Preliminary testing was therefore abandoned and the wet samples were sent off to the local laboratory. They agreed to test pH prior to ICPMS, but testing Nitrate or similar compounds would require iron chromatography or similar tests at a substantially higher cost per sample.
Many elements that were included in the ICPMS results were below the test’s detection threshold of 0.5 ppb for all salt samples. Accordingly, these elements are not included in the data and comparisons below. These elements were: Beryllium, Scandium, Gallium, Germanium, Yttrium, Zirconium, Niobium, Ruthenium, Rhodium, Cadmium, Tin, Cesium, Lanthanum, Cerium, Praseodymium, Neodymium, Samarium, Europium, Gadolinium, Terbium, Dysprosium, Holmium, Erbium, Thulium, Ytterbium, Lutetium, Hafnium, Rhenium, Osmium, Iridium, Platinum, Thallium, Thorium, and Uranium.
Silver and Tellurium were below the test’s 5 ppb detection limit for all salts. Interestingly, Silver levels for Instant Ocean and Coralife were reported at 248 ppb and 410 ppb respectively in the 1999 Atkinson and Bingman study.
Gold and Mercury were below their 50 ppb detection limit, and Iron was below the 500 ppb detection limit for all samples.
The tests for several elements showed little significant difference between salt mixes. The data for these elements can be found in the Appendix.
Natural Sea Water (NSW) levels were taken from a variety of reports and studies. The range of values for NSW element concentrations varies depending on the study and the area of the ocean that was sampled. For all elements we list both the lowest and highest values to establish an acceptable range.
pH
Natural sea water averages pH 8.25. Saltwater aquaria normally range from 7.8 to 8.5. The measured pH for each mixed salt sample is shown below. The pH measurement was made 24 hours after the initial mix and after regular aeration.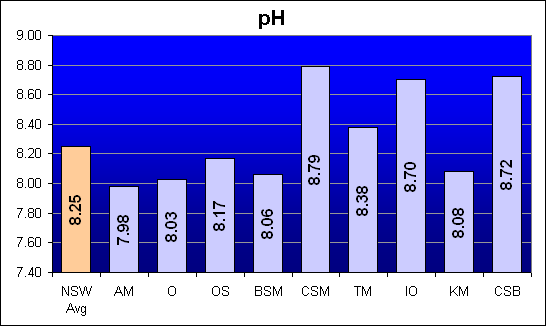
Aluminum (ppb)
This is one of the elements called out in Dr. Shimek’s study as showing significantly higher toxicity levels for Instant Ocean and Coralife as compared to Bio-Sea MarineMix and Crystal Sea Bio-Assay. As can be seen, Bio-Sea MarineMix tested with the second highest Aluminum concentration and Crystal Sea Bio-Assay was comparable to Instant Ocean.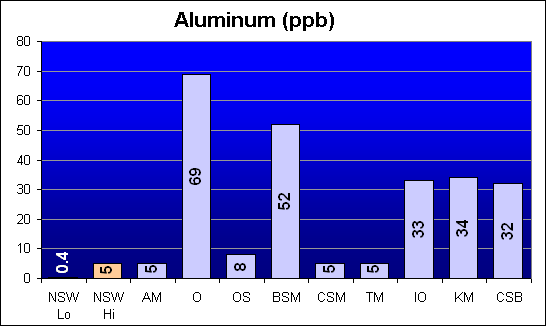
Antimony (ppb)
Antimony is a potential heavy metal toxin that was not called out in Dr. Shimek’s study. The two salts from Marine Enterprises: Crystal Sea MarineMix and Crystal Sea Bio-Assay, along with Kent Marine, had significantly higher than NSW levels (about ten times NSW).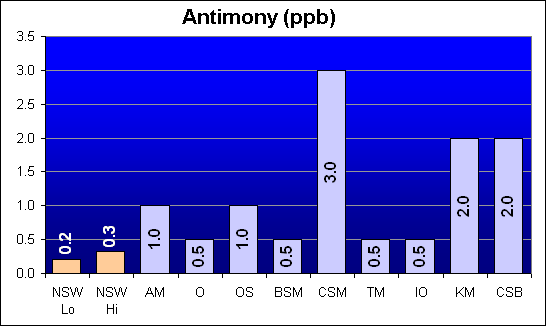
Boron (ppm)
Boron is a not considered a heavy metal. It is a semiconductor and is lighter than Carbon, Nitrogen, and Oxygen. It is normally found in seawater as Borate ions. There is a wide variation in Boron levels in the salt mixes tested, from less than one thirtieth of NSW values to more than three times NSW. Interestingly, Crystal Sea Bio-Assay had the second highest Boron level, Crystal Sea MarineMix, from the same manufacturer, was the highest, and Instant Ocean was the closest to NSW levels.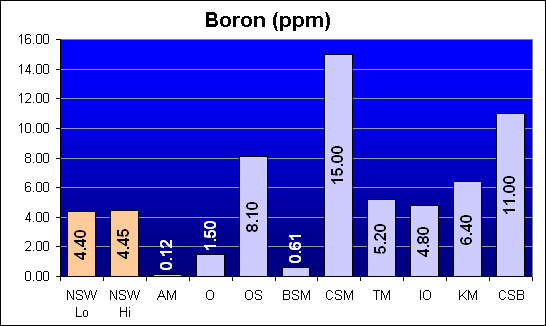
Barium (ppb)
Barium is another potential heavy metal contaminant. Both Bio-Sea MarineMix and Crystal Sea Bio-Assay are similar to NSW. Instant Ocean tested with the lowest Barium concentration.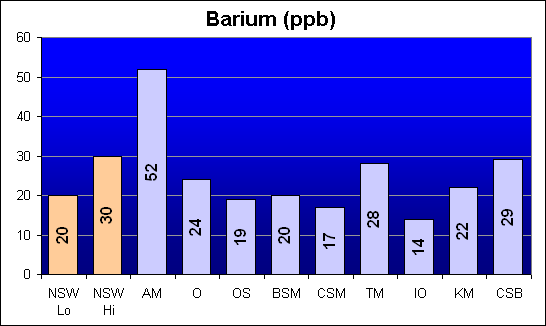
Bismuth (ppb)
Bismuth is a potential heavy metal contaminant that is not called out in Dr. Shimek’s study. All salt mixes except AquaMedic tested below the 5 ppb detection limit of the test.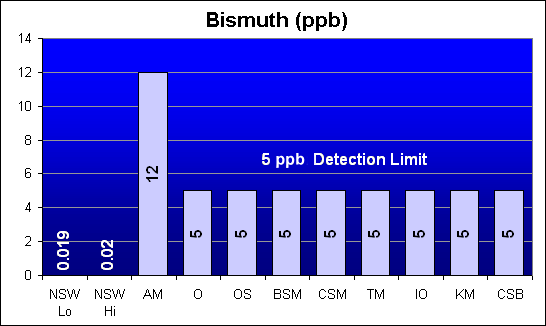
Bromine (ppm)
Bromine is a halogen, a non-metallic salt-forming element related to Chlorine, Fluorine, Iodine, and Astatine. It is present in sea water mostly as free bromide ions. All salt mixes have excess levels of Bromine compared to NSW, but it should not be a toxic contaminant at any of these levels.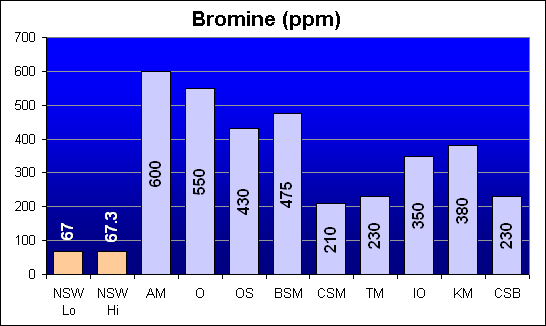
Cobalt (ppb)
A potential metal toxin, Cobalt levels tested below the 0.5 ppb test detection threshold for Instant Ocean, Kent Marine and Crystal Sea Bio-Assay. All other salt mixes were significantly higher than NSW levels, including Bio-Sea MarineMix.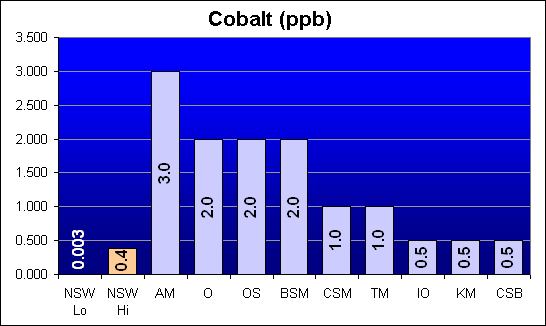
Chromium (ppb)
Chromium is another element called out in Dr. Shimek’s study as showing significantly higher toxicity levels for Instant Ocean and Coralife as compared to Bio-Sea MarineMix and Crystal Sea Bio-Assay. Our test results show that, while all salts were higher than NSW, Instant Ocean was actually the lowest. Bio-Sea MarineMix was the second highest, and Crystal Sea Bio-Assay had the third highest Chromium concentration.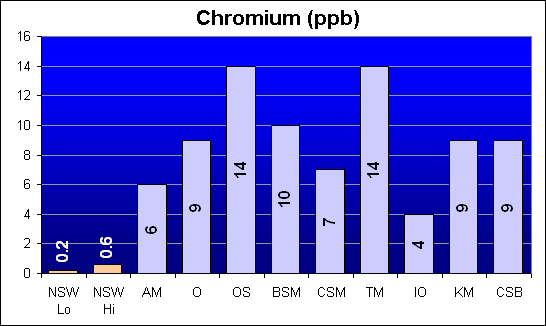
Copper (ppb)
Another heavy metal toxin, Copper levels were higher than NSW in all salt mixes. Once again, Dr. Shimek’s study indicates lower levels for Bio-Sea MarineMix and Crystal Sea Bio-Assay than for Instant Ocean and Coralife. In fact, Instant Ocean tested with the second lowest Copper concentration. The other two salts in his study were roughly in the middle of the field.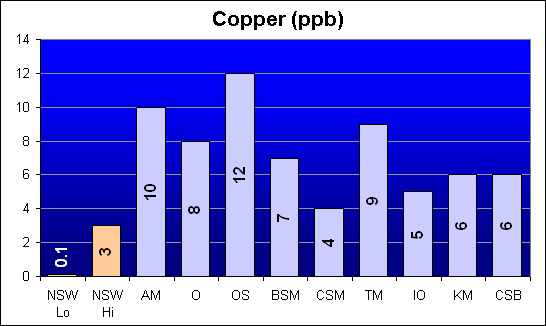
Lithium (ppm)
Lithium is a lightweight metal that exists in sea water as a free positive ion. Lithium levels were below or in the general range of NSW levels for all salts except those from Marine Enterprises: Crystal Sea MarineMix and Crystal Sea Bio-Assay. Those two salt mixes were approximately 70 times NSW levels.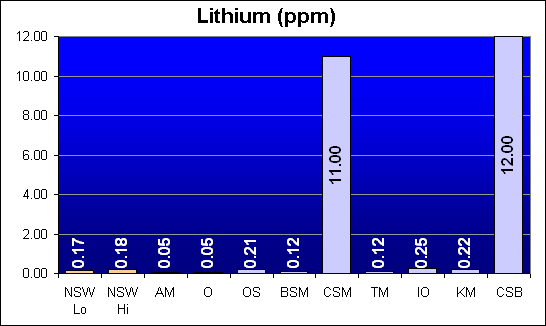
Manganese (ppb)
Manganese is another potentially toxic element which was reported in Dr. Shimek’s study to be significant higher in Instant Ocean and Coralife than in Bio-Sea MarineMix or Crystal Sea Bio-Assay. Our results show that, while all salt mixes were higher than NSW, Instant Ocean was comparable to the other salts. Only Oceanic showed a significantly high level – about ten times the other salt mixes.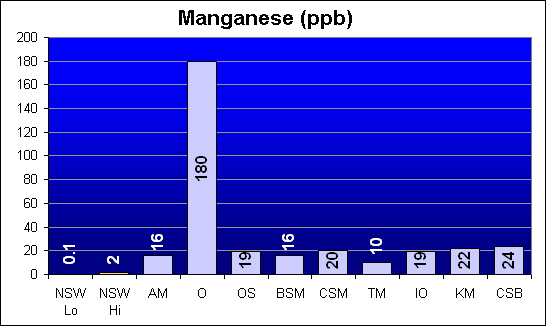
Molybdenum (ppb)
Once again, Dr. Shimek’s study showed much higher levels in Instant Ocean and Coralife than in Bio-Sea MarineMix or Crystal Sea Bio-Assay. However, our results show that Instant Ocean and Bio-Sea MarineMix both have the same low level and Crystal Sea Bio-Assay has the second highest Molybdenum concentration.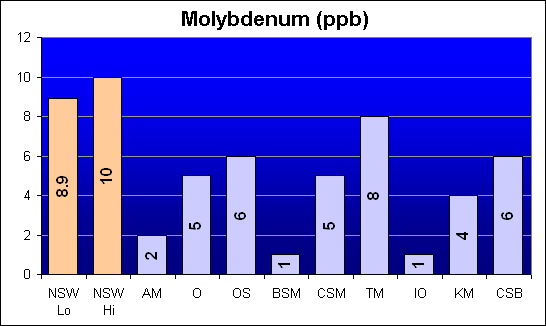
Nickel (ppb)
Nickel is another metal that was reported in Dr. Shimek’s study to be significant higher in Instant Ocean and Coralife than in Bio-Sea MarineMix or Crystal Sea Bio-Assay. In fact, Crystal Sea Bio-Assay had the highest Nickel concentration and Instant Ocean was the third lowest.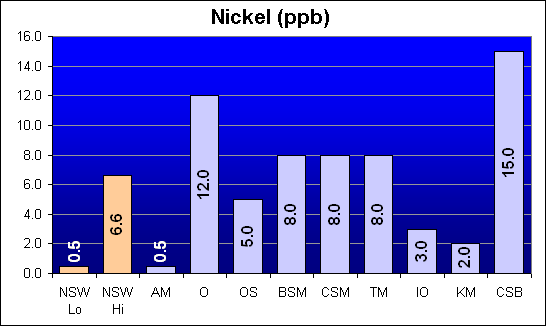
Lead (ppb)
Lead is a toxic heavy metal that was reported in Dr. Shimek’s study to be over 100 times higher in Instant Ocean than in Crystal Sea Bio-Assay. Our results show that all salt mixes were higher than NSW, but Crystal Sea Bio-Assay was in fact slightly higher in Lead levels than Instant Ocean. Bio-Sea MarineMix had the same level as Instant Ocean.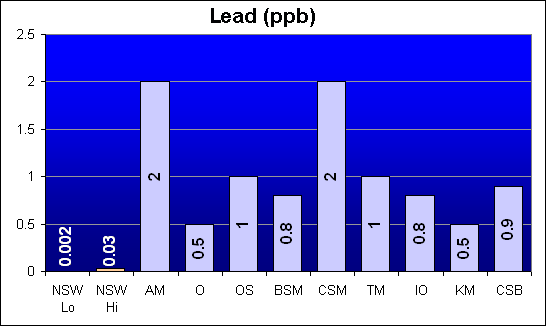
Palladium (ppb)
Palladium was not specifically called out in Dr. Shimek’s study. All salt mixes were about 100,000 times higher than NSW, but similar to each other within a factor of ten.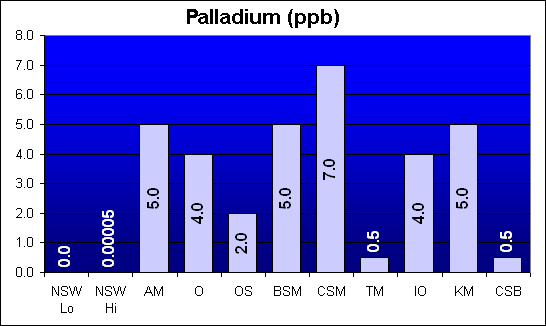
Selenium (ppb)
Selenium is another potential heavy metal toxin that was not called out in Dr. Shimek’s study. The salts from Marine Enterprises tested lower than other salts for this element. However, all salt mixes were 100 to 400 times the average NSW level.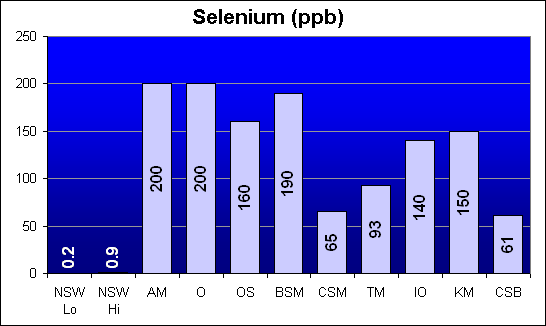
Silicon (ppm)
All salts except Crystal Sea Bio-Assay tested below the minimum detection limit of the test.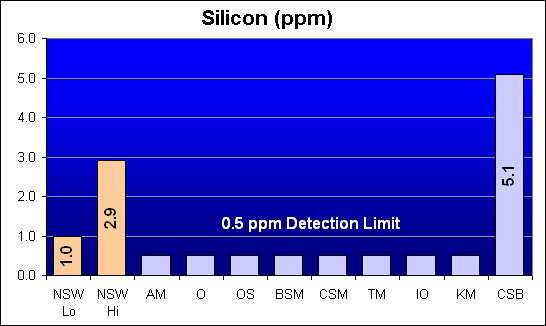
Strontium (ppm)
Strontium is an essential element for stony coral growth. Oceanic and Crystal Sea MarineMix showed deficient levels of Strontium, whereas Omega Sea tested at roughly double NSW levels. All other salt mixes were similar to NSW.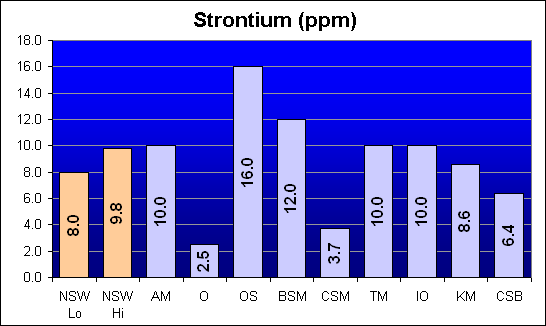
Titanium (ppb)
Titanium is yet another element that was reported in Dr. Shimek’s study to be significantly higher in Instant Ocean and Coralife than in Crystal Sea Bio-Assy. Our results show that four of the salt mixes are roughly eight times NSW level: AquaMedic, Crystal Sea MarineMix, Instant Ocean, and Crystal Sea Bio-Assay. All other salts were two to three times NSW.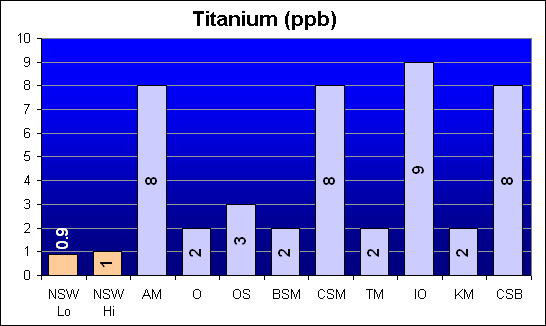
Zinc (ppb)
Zinc was also reported in Dr. Shimek’s study as being several times higher in Instant Ocean and Coralife than Crystal Sea Bio-Assay or Bio-Sea MarineMix. We show that Oceanic has a significantly higher than NSW level of Zinc, and that AquaMedic, Omega Sea and Tropic Marin are about five times the average NSW level. Other salts are at or below NSW levels.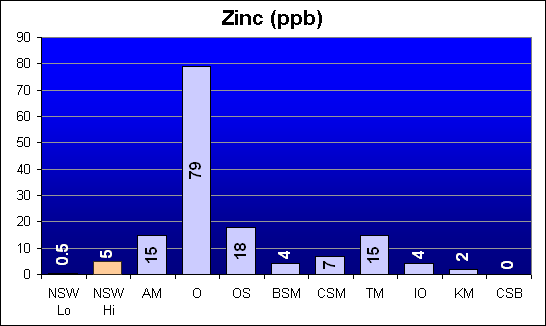
Summary
There are three separate areas to be addressed:
- Does the manufacturer’s data hold up to close scrutiny?
- Are there heavy metal concentrations revealed in these tests that could account for Dr. Shimek’s bio-assay results?
- Do these results shed any light on the salt mix choices available to the hobbyist?
These items will be discussed in Part II of this article next month.
References
- Atkinson, M. and C. Bingman 1999. The Composition of Several Synthetic Seawater Mixes. Aquarium FrontiersOnline. March, 1999
- Inland Reef and Ron Shimek 2001. Analysis of TLF Combisan. Reefs.org online article library (http://www.reefs.org/library/article/combisan_analysis.html).
- Shimek, R. L. 2003. The Toxicity of Some Freshly Mixed Artificial Sea Water; A Bad Beginning For A Reef Aquarium. Reefkeeping.com. Volume 2. Number 3. March, 2003.
- Randy Holmes Farley 1998. Understanding Seawater. Aquarium Frontiers Online. July, 1998
End Notes
1 “This study has demonstrated that the artificial sea water made using some common and popular commercial artificial salt water mixes is toxic to sea urchin larvae using a variant of a standard bioassay. Such water will also likely have effects on other animals.” Shimek, 2003
2 “This study also showed that some artificial sea water mixes produced water that could support larval development as well as could natural seawater.” Shimek, 2003
3 “The two salts that made artificial seawater with the lowest survivorship of larvae consistently have heavy metals concentrations hundreds to hundreds of thousands times those found in natural seawater. Those salts that had the best survival had heavy metals concentrations that generally ranged about from, at worst, about one third to, at best, one thousandth those values.” Shimek, 2003
4 “… I had to rely on data provided by the manufacturer of Crystal Sea Marinemix Bioassay Formula. For the Bio-Sea Marinemix, I used the data from one of the advertising brochures describing the salt.” Shimek, 2003
5 “Fortunately, there is no a priori reason to doubt the veracity of either of these sources.” Shimek, 2003



0 Comments