
For its time, my first saltwater aquarium was state of the art. It was a 20-gallon all glass tank held together with ‘space age’ silicone glue. Its filtration system consisted of air-driven sub-sand filters. It was my aim to keep its two inhabitants (a Beau Gregory damselfish – Stegastes leucostictus – and a Condylactis anemone) as happy as possible (which meant alive, given my inexperience and limited resources.) It was a big challenge for a 9-year old back in 1964. My mentor, Mrs. Perry at Cobb Pets, advised me to check the water’s specific gravity and pH. Specific gravity was easy enough (just float a hydrometer in the tank and keep a marking on its neck at a certain level by adding freshwater) but pH was a little more complicated. This parameter was checked by adding a colored liquid to a vial containing a sample of aquarium water. As if by magic, the sample’s color changed and was then checked against a comparison chart consisting of a series of colored squares. The results of my first test directed me to add baking soda to raise the pH. I dutifully did so and – no change. I continued this process until I had added the whole box of baking soda.
I will never know exactly what caused the death of the fish and anemone but it happened immediately after this episode. As bad as it was for my pets, it was devastating to me. All that work to earn my one dollar-a-week allowance was down the tubes. Worse, I was responsible for their deaths. I buried their remains on the fern-covered bank of the creek that ran across our property. I now believe the liquid reagent had probably passed its expiration date and had produced erroneous results. It was a hard-learned lesson.
Things haven’t changed much over the years. Failure to understand the importance of this critical parameter and how to test for it, correctly interpret the results, and take appropriate actions can and will result in disaster. What has changed dramatically is the availability and affordability of methods and instruments now routinely available to hobbyists. This article will review some of these and examine the pros and cons of each.
Definition of pH
pH is the measure of the logarithm of the Negative logarithm of the hydrogen ion activity. In plain English, it is an estimation of a substance’s acidic or basic nature as expressed on a scale of 0 to 14, with 0 being very acidic and 14 very basic. Neutral (neither acidic or basic) is at 7 on this scale. Hydrogen ions are predominant at acidic pH values, while hydroxyl ions being in the majority indicate a basic nature.
pH is not ‘acidity’ or ‘alkalinity’ – these parameters measure the resistance to changes in pH and are tested for by separate means. Depending upon the reference, pH stands for ‘potential of hydrogen’ or the French term for “pouvoir hydrogène” meaning hydrogen power.
Importance of Measuring pH
pH is a characteristic of liquids (for our purposes) that affects their chemical makeup, such as solubility of nutrients (good, as long as we don’t go overboard.) Low pH can make potentially toxic heavy metals become soluble. Enzyme activity is affected by pH (these have preferred pH ranges.) High pH can dissolve cellular lipid membranes. Aquatic organisms have preferred pH ranges as well. For a quick glimpse of pH values seen in various environments of interest to aquarists, see Table 1.
| Source | pH |
|---|---|
| Rio Negro River | 5.1 |
| Rainwater | 5.6 |
| Amazon (white water) | 6.9 |
| Pure Water | 7 |
| Seawater | 8.2 |
| Lake Tanganyika (surface) | 9 |
Measuring pH
There are several ways to estimate pH. Each has advantages and disadvantages. We’ll begin our examination of these with the least expensive:
Litmus Paper
Litmus is a material obtained from lichens (its name originates from the Old Norse word litmosi meaning ‘dye’ and ‘moss.’) This litmus derivative changes color in a predictable manner when exposed to different pH values. This sensitivity makes it an easy and inexpensive way to estimate pH. Litmus paper is that to which these water-soluble dyes have been added and color changes caused by immersion in a sample can indicate if it is acidic or basic in nature. Its working pH range is about 5 to 8. Examination of color changes should be done under full spectrum light.
Advantages:Inexpensive (~$5 US or so.) Quick, easy to perform.
Disadvantages: Yields ‘ballpark’ values. Subject to influence by color of the sample, reductants and oxidizers. Interpretation of result requires visual acuity. Reagent has limited shelf life.
Indicator Dyes
There are quite a few of these pH indicators. These can be purchased in either powder or liquid form. Usually used in analyses involving titration. I’ve listed the qualities of several:

Figure 3. This pH test kit by API uses meta-cresol purple as the indicator. Examination of color changes are best done with natural light and against a white background.
- Phenolphthalein: An acid/base indicator which is colorless in an acidic solution and pink to red in a basic one. Range of ~8.3 to 10.
- Methyl Orange: Changes from yellow to red at a pH of about 3.7.
- meta-Cresol Purple: Orange-yellow at 7.4 and turning purple at higher pH (up to about 8.8.)
- Bromothymol Blue: Blue at 7.5, greenish at ~6.2 to 6.8, and yellow at about 6.
- Universal Indicator: A mix of several indicators which allows a broad range of pH estimations.
Advantages: Relatively inexpensive (~$10 US.) Some dyes allow other analyses (such as alkalinity) without use of a pH electrode when using reagent for that purpose.
Disadvantages: Same as those for litmus paper. Single dyes have limited pH ranges. Results can be influenced by turbidity and/or color of the liquid tested. Comparisons should be done against a white background under full spectrum light. Reagents have a limited shelf life and should be marked with an expiration date.
pH Electrodes
As difficult as it might be for newcomers to the hobby to imagine, use of pH electrodes by hobbyists outside of Europe was practically unheard of about 30 years ago. This changed in the 1980’s when a German company (Dupla GmbH) began exporting advanced instrumentation to North America. Today, pH meters are used as a matter of routine. Availability and competition has resulted in meters with very affordable price tags.
A pH electrode is a selective probe for the hydrogen ion (H+). pH electrodes actually use two electrodes, a sensing electrode and a reference electrode. Most of the time these electrodes are combined in a single electrode body. The sensing electrode incorporates a thin layer of hydrogen-sensitive glass at the end of the electrode body. Voltage changes at the sensing electrode as a function of hydrogen ion activity (e.g. voltage increases in acidic solution and decreases in basic solutions). The reference electrode supplies a constant voltage that we use to determine the difference between the sensing electrode. The net mV response is sent to the meter, where it can be converted to a pH value.
Probe Anatomy and Terminology
To understand how a pH electrode works, we should understand some of the terms used to describe its construction, as well as other terms.
Body: A hollow tube containing the working components of a pH electrode. They can be made of glass or a chemical-resistant plastic, such as PEI.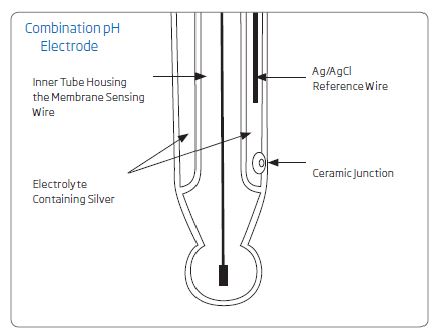
Buffer: For our purposes, a standard solution demonstrating an acidic, neutral, or basic pH and used to calibrate a pH meter. Some buffers are color coded for ease of recognition.
Calibration: The act of checking or rectifying the graduation of an analytical device.
Junction: Where two things come together, in this case, the material being tested and reference internal solution. Junctions can be made of many materials if it is porous and allows the reference solution to pass through it. Ceramic, cloth, etc. are commonly used. There are electrodes with single, double, or annular junctions.
Frit: A partially fused glass or ceramic material sometimes used as a junction.
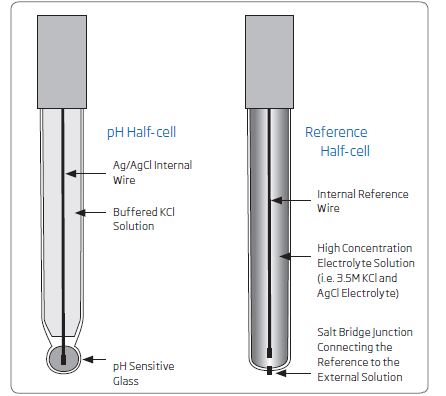
Figure 4. Internal components of a pH electrode. Not shown for clarity is a protective shroud surrounding the fragile glass bulb. The junction is on the side of some pH electrodes
ATC: Automatic Temperature Compensation. Since the pH of a solution is temperature dependent, ATC corrects for temperature’s effect. ATC requires a temperature sensor, sometimes built in to the electrode near the glass bulb.
Reference electrode: An electrode that supplies a known, constant voltage, usually made from a silver-silver chloride wire and filled with buffered electrolyte.
Sensing electrode: A silver/silver chloride wire in a tube with a pH-sensitive glass bulb at the end.
Types of pH Electrodes
There are several types of electrodes. Some, generally older, electrodes (seldom seen today in my experience) consist of two separate bodies. Today most are a combination probe where the anode and cathode are housed separately within a single body. The shape of the glass bulb often determines what the electrode will measure. Spherical bulbs, with their large surface area, are good for general purpose measurements in aqueous environments. Conical bulbs can pierce semi-solids (such as meats and other foods) and soils. Flat glass ‘bulbs’ can be used to measure pH of skins, leather, etc. Some electrodes are refillable while others are not and are filled with chemical gels. Some have replaceable junctions or probes.

Figure 5. The affordable pH Checker from Hanna Instruments.
A Quick Overview of pH Meters
Our review of pH meters is of those manufactured by Hanna Instruments (Woonsocket, Rhode Island, USA.) Hanna has been in business since 1978, and today offers over 3,000 products to its customers worldwide. Many of their products are of interest to aquarists.
All Hanna pH meters reviewed here come with calibration buffers, electrode cleaning solution, and carrying case. We’ll begin our review with:
pH Checker (HI98103)
The HI98103 Checker® entry level pH meter would be a valuable addition to many aquarists’ toolbox. It offers 0.1 pH unit resolution at an affordable price. Affordability comes at a price as the device offers only two calibration points (pH 4.01 and 7.01 or 7.01 and 10.01) without automatic temperature compensation (ATC) or temperature measurement capability. Since it is generally recommended that calibration points bracket the sample’s expected pH, this meter is best suited for freshwater tanks mimicking acidic biotopes such as the Amazon River (although it is certainly capable of measuring pH values normally seen in reef and African cichlid aquaria albeit with slightly reduced accuracy due to the two calibration points.) The electrode is replaceable and the junction is made of paper.
- Range: 0 to 14 Units
- Resolution: 0.1 Units
- Accuracy: ±0.2 Units
- Calibration Points: Two; pH 4.01, 7.01 or 10.01
- Automatic Temperature Compensation: No
- Temperature Measurement/Display: No
- Replaceable Probe: Yes
- Electrode Diameter: 8 mm (~5/16″)
- LCD Size: 3/8″ (~10mm)
- Battery: 1-CR2032; approximately 1,000 hours battery life.
- Suggested Retail Price: $29.50 US
pHep pH and Temperature Tester (HI98107)
The HI98107 pHep pH and temperature tester is a more advanced version of the pH Checker (described above.) In addition to pH determinations in the range of just about any aquarium – Amazon to Reef – it also measures temperature with Automatic Temperature Compensation (ATC.) It includes two calibration buffers (4.01 and 7.01) with a third available (10.01, which is recommended for reef aquaria.) The junction is made of paper. The electrode is not replaceable.
- Range: 0 to 14 Units
- Resolution: 0.1 Units
- Accuracy: ±0.1 Units
- Calibration Points: Three; pH 4.01, 7.01, and 10.01 (4.01 & 10.01 are supplied)
- Automatic Temperature Compensation: Yes
- Temperature Display: Yes, can be set to °F or °C, with ±0.5°C accuracy.
- Replaceable Probe: Yes
- LCD Size: 0.3125″ or ~8 mm
- Battery: 1-CR2032; approximately 800 hours battery life.
- Suggested Retail Price: $39.50 US
pHep5 Waterproof pH and Temperature Tester (HI98128)
The HI98128 pHep 5 pH meter is the most advanced of any of Hanna’s pocket pH meters. It offers resolution of 0.01 units with an accuracy of ±0.05 and offers ATC. It is waterproof and floats. This device offers flexibility in critical measurements as it can recognize 5 different calibration buffers.
- Range: -2 to 16 Units
- Resolution: 0.01 Units
- Accuracy: ±0.05 Units
- Calibration Points: Two sets: 4.01, 7.01, 10.01 or 6.86, 9.18.
- Automatic Temperature Compensation: Yes
- Temperature Display: Yes, can be set to °F or °C, with ±0.5°C accuracy.
- Replaceable Probe: Yes
- LCD Size: 0.3125″ or ~8 mm (character size)
- Battery: 4-1.5v batteries; approximately 300 hours battery life.
- Suggested Retail Price: $98.00 US
HALO Wireless Field pH Meter (HI12302)
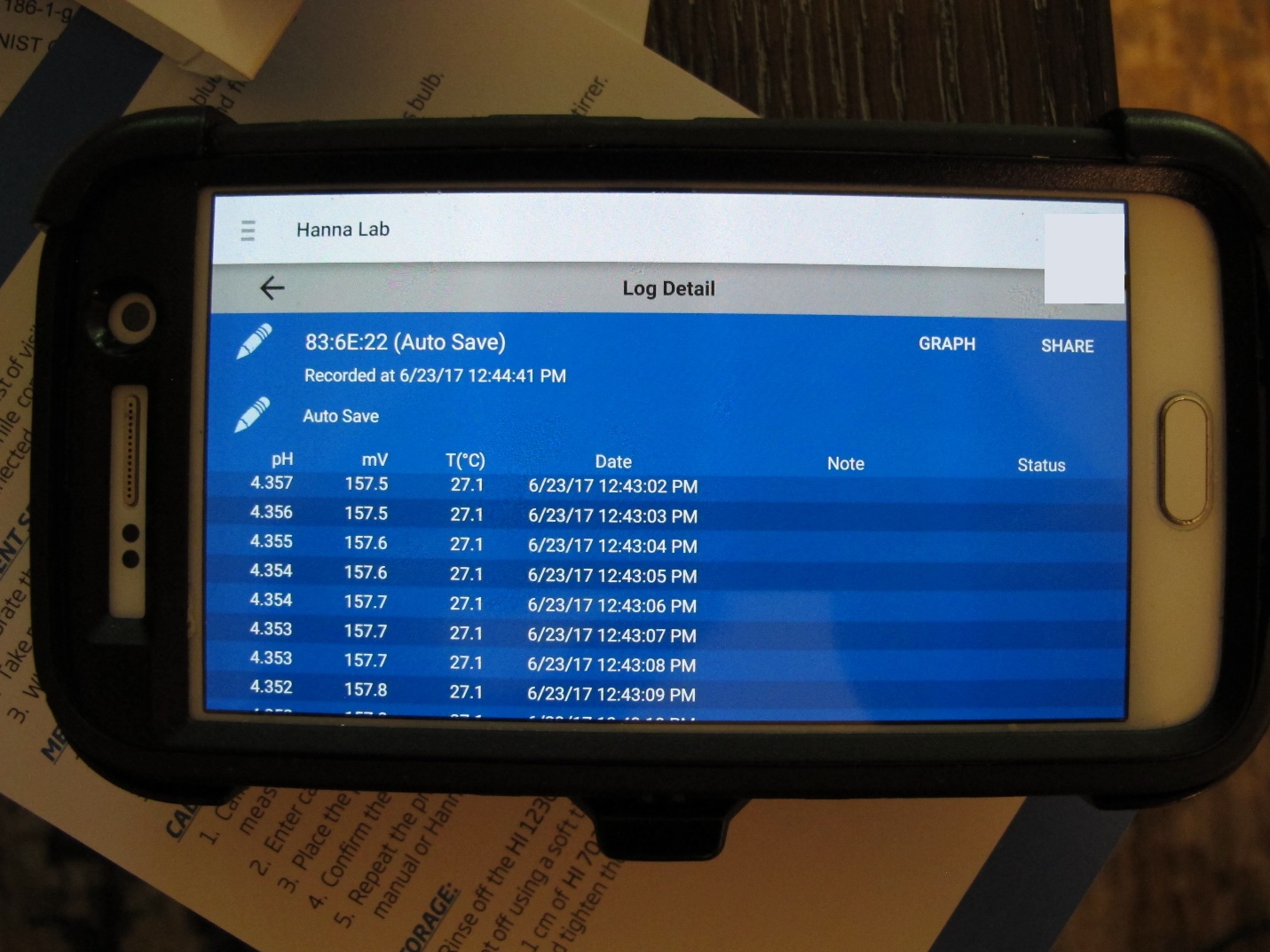
The HI12302 Halo Field pH Meter is an interesting device that offers many options. First and foremost, it is a wireless pH electrode that can be operated via Bluetooth from Android or iOS devices. Those who feel technically-challenged should have no fear. I found the setup incredibly simple. I went to Hanna Instruments website, clicked on the HALO link, and downloaded the app for my smart phone. Once downloaded (it’s free, and took about 2 minutes), I opened the app and the software recognized my HALO pH electrode. From there, it is only matter of clicking the appropriate icons to calibrate the electrode, graph data, view probe data, etc. I honestly believe that the procedure could have not have been easier. Software records pH and temperature once per second. The data log information shows electrode ID number, calibration date, calibration points, calibration slope, date and time of measurements, pH, temperature, millivolts, and so on (See Figures 9-11).
Tip options include spherical (general purpose and aqueous), conical (foods, semisolids, soils, etc.) and flat tip (skin, paper, leather, etc.) The HALOs polyetherimide (PEI) plastic body approved for food contact and is impervious to anything the reefkeeper can throw at it (unless you’re completely unhinged and dose your tank with aromatic hydrocarbons and/or partially halogenated solvents.)
- Range: 0 to 14 Units
- Resolution: Selected by user: 0.1, 0.01 or .001 Units
- Accuracy: ±0.005 Units
- Calibration Points: Seven; pH 1.68, 4.01, 6.86, 7.01, 91.8, 10.01 and 12.45.
- Automatic Temperature Compensation: Yes
- Replaceable Probe: No
- Electrode Diameter: 12 mm (~1/2″)
- Data Logging: Yes
- Battery: Lithium battery, 500 hours.
- Suggested Retail Price: $160 US
See here to see if your phone or tablet is compatible with HALO’s technology: http://hannainst.com/halo
For further information on these and other Hanna Instruments products, see here: http://hannainst.com
All Hanna probes and electrodes are covered by a 6-month warranty.

Figure 10-11. In data logging mode, pH measurements made by the HALO electrode can be viewed in tabular form or viewed as a graph. Annotations are possible, and data can be exported to an Excel spreadsheet.
Other Considerations
I’ll now briefly cover other things you should know about when purchasing a meter or electrode.
Connectors
Those pH devices having separate electrodes must be connected to the meter via a connector (unless it is a wireless device such as Hanna’s HALO.) Although this might seem trivial, it can have long term and potentially expensive consequences. Some manufacturers use specialized connectors thus ensuring continued use and purchase of their electrode. Perhaps the most common is the Bayonet Neill-Concelman (BNC) quick-connector/disconnector. Less commonly seen is the US connector. Some European meters use a S7 connector.
Junctions
A junction in a pH electrode is the meeting point of two worlds – the probe’s internal solution and the sample to be tested. There are some specialized terms used when describing junctions, their composition, and geometry. As mentioned, junctions allow an electrode’s reference solution to flow into the sample being tested. As such, they are subject to clogging, especially in oily samples, or those with high protein content, or suspensions. Some electrodes use a junction made of cloth. More expensive electrodes use porous ceramic materials. Some junctions are made of the plastic PTFE (polytetrafluoroethylene) and are designed for harsh environments, including those with high hydrocarbon content. PTFE junctions are sometimes relatively large and form an annular ring around the glass bulb (ceramic junctions are often small wicks only 1 millimeter or so in diameter.) All junctions are subject to clogging.
Fortunately, for reef hobbyists, a general purpose pH probe with cloth or ceramic junctions are satisfactory.
Cleaning pH Electrodes
Always remember electrodes are scientific instruments that should be treated with care. Although the plastic body is rugged, the glass bulb is fragile and careless treatment will result in its breakage. Electrodes used only seldomly will naturally enough require infrequent cleaning; however, if your electrode is continuously immersed in organic soups (such as the water in some aquaria) the hobbyist is well advised to implement a routine cleaning schedule. It is entirely possible for a sensor to become coated with biological growths and proteins. Foods (and catastrophic submersible pump failures) could add oils to an aquarium’s water and result in a fouled electrode. Fortunately, cleaning solutions can help maintain an electrode’s health. Follow the manufacturer’s directions. Don’t rub the electrode – always blot it dry to prevent potential of static charges.
Fillable v. Non-fillable Gel Electrodes
Some electrodes can be refilled with solutions specifically tailored for their use, while others are filled with a gel. Generally, gel probes have a slower response to changes in pH. Most probes for use in aquaria are gel filled.
Calibration
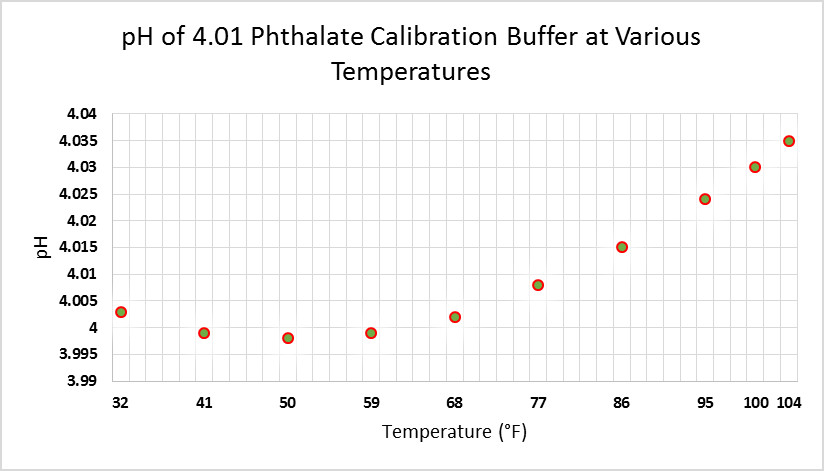
Correct calibration of the pH electrode is essential to obtaining correct results. This is easier when the device has automatic temperature compensation (ATC.) See Figures 12-14 for examples of effects of temperature on calibration standards.
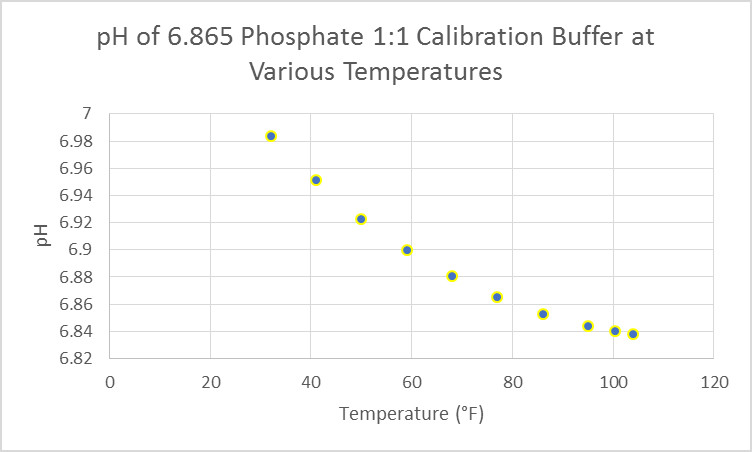
Figure 13. Temperature affects the pH of this potassium dihydrogen phosphate/disodium hydrogen phosphate buffer (6.865.) Fortunately, calibrations made at room temperature are very close when using a device without ATC.
Proper calibration of a pH electrode requires a little patience and attention to detail. New probes should be properly hydrated (see directions with your device.) Although single point calibration is possible, it is better to calibrate to 2 points (which should bracket the expected pH. For reef aquaria, use 7.01 and 9 or 10 buffers. Be aware some meters have auto-recognition of buffers and require use of specific solutions. Before calibration, inspect the electrode for any damage (especially the glass bulb.) There should be no biological growths on the glass bulb. If there are present, use an electrode cleaning solution recommended by the manufacturer. Proper cleaning will remove bio-growths, oils, protein contamination, etc. The electrode, if refillable, should be full of the solution recommended by the manufacturer. Once satisfied the electrode is clean and in good condition, place the electrode in the first calibration solution. Make sure the electrode’s glass bulb and junction are completely submerged in the calibration solution (I use a 30-milliliter beaker where 7 milliliters of buffer is sufficient for calibration purposes.) Vigorously stir the solution using the electrode (if a magnetic stirrer is not available) and allow time for temperature of the electrode and solution to equalize. Enter the calibration into the device’s memory (usually by the press of a button while in calibration mode.) Rinse the electrode with distilled water and blot it dry with tissue paper (preferably a ‘lab grade’ tissue such as Kimwipes.) NEVER rub the electrode with the paper – it is possible to create a static charge that can affect calibration and hence measurements. Calibration is now complete if performing a single point calibration. If not, repeat the procedure for a 2- or 3-point calibration. When measuring the pH of a sample, stir the solution manually or with a stirrer, and allow time for temperature compensation. Good lab practice dictates that pH and temperature are recorded.
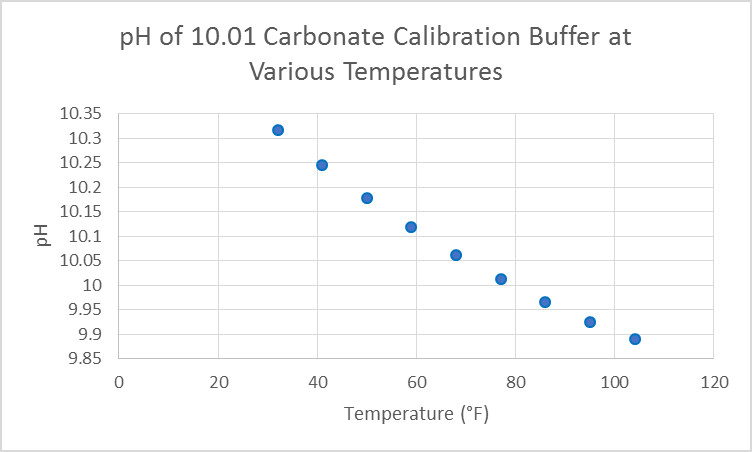
Figure 14. This buffer’s pH (sodium bicarbonate/sodium carbonate) can be affected by temperature (another case for using a device with ATC.) Atmospheric carbon dioxide can affect it over a period of time.
Degradation of Calibration Buffers
As with most chemical reagents, pH buffers are subject to degradation over time. Some buffers are formulated to resist changes and can have extended shelf life (years.) Look for buffers that have an expiration date marked on the container. Carbonate buffers generally have a shelf life less than those of neutral and acidic nature due to the effects of atmospheric carbon dioxide. Discard buffers that have contacted the electrode during the calibration process. Also discard buffers if mold is noted growing in them (usually buffers in the range of 4 or so.) Do not use buffers to adjust the pH of your aquarium.
pH Electrode Storage
Storage of a pH probe must be done properly. Mostly importantly, the glass bulb must remain hydrated. Second, the storage solution should be formulated to prevent osmosis between it and the electrode’s internal solution/gel. It should also contain an anti-microbial agent to prevent mold and other growths.
For necessary pH calibration buffers, storage solutions and accessories see here: http://hannainst.com/ph-solutions







0 Comments